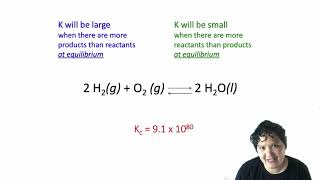16. Chemical Equilibrium
Reaction Quotient

Get help from an AI Tutor
Ask a question to get started.
Problem 5
Textbook Question
Textbook QuestionThe reaction A21g2 + B21g2 ∆ 2 AB1g2 has an equilib- rium constant Kc = 9. The following figure represents a reaction mixture that contains A2 molecules (red), B2 mol- ecules (blue), and AB molecules. What statement about the mixture is true? (LO 15.5)

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
189
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos