13. Liquids, Solids & Intermolecular Forces
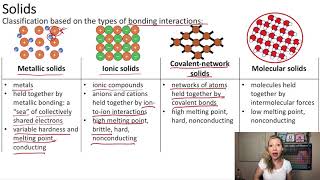
Atomic, Ionic, and Molecular Solids

Get help from an AI Tutor
Ask a question to get started.
Problem 101
Textbook Question
Textbook QuestionWhich statement correctly describes a difference between graphene and graphite? (a) Graphene is a molecule but graphite is not. (b) Graphene is a single sheet of carbon atoms and graphite contains many, and larger, sheets of carbon atoms. (c) Graphene is an insulator but graphite is a metal. (d) Graphite is pure carbon but graphene is not. (e) The carbons are sp2 hybridized in graphene but sp3 hybridized in graphite.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
614
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos