10. Periodic Properties of the Elements
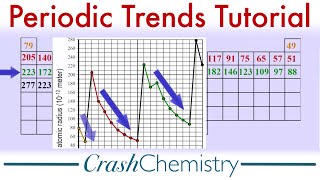
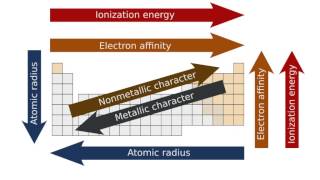
Periodic Trend: Cumulative

Get help from an AI Tutor
Ask a question to get started.
Multiple Choice
Multiple ChoiceFor main-group elements, the atomic radius increases going down a group because:
A
Effective nuclear charge decreases.
B
The mass of an element increases.
C
The highest principal quantum number of the valence orbitals increases.
D
Electron affinity increases.
E
Both effective nuclear charge and the principal quantum number of valence orbitals increases.
580
views
1
rank
1
comments
Related Videos
Related Practice
Showing 1 of 8 videos