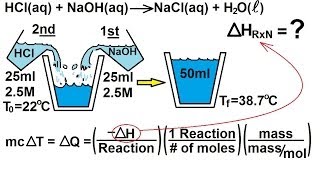
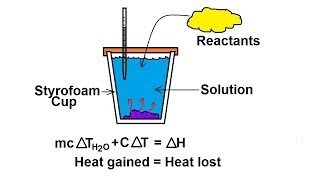
8. Thermochemistry
Constant-Pressure Calorimetry

Get help from an AI Tutor
Ask a question to get started.
Multiple Choice
Multiple ChoiceA 115.6 g piece of copper metal at 182.5 ºC is placed into 120.0 mL of methylene chloride at 31.0 ºC within a coffee-cup calorimeter. If the final temperature of the solution is 50.3 ºC, what is the specific heat of methylene chloride? Assume the calorimeter absorbs a negligible amount of heat. The specific heat of copper is 4.184 J/g ∙ ºC and the density of methylene chloride is 1.33 g/cm3.
A
5.022 J/g°C
B
8.178 J/g°C
C
12.94 J/g°C
D
17.28 J/g°C
E
20.76 J/g°C
645
views
3
comments
Related Videos
Related Practice
Showing 1 of 11 videos