9. Quantum Mechanics
Bohr Model

Get help from an AI Tutor
Ask a question to get started.
Problem 36
Textbook Question
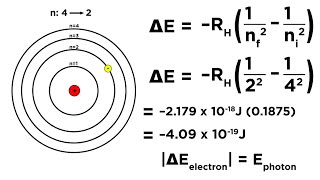
Textbook QuestionClassify each of the following statements as either true or false: (a) A hydrogen atom in the n = 3 state can emit light at only two specific wavelengths (b) a hydrogen atom in the n = 2 state is at a lower energy than one in the n = 1 state (c) the energy of an emitted photon equals the energy difference of the two states involved in the emission.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
492
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos