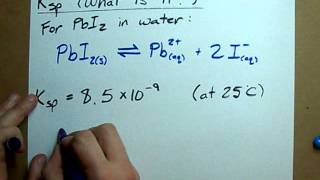18. Aqueous Equilibrium
Solubility Product Constant: Ksp

Get help from an AI Tutor
Ask a question to get started.
Problem 14
Textbook Question
Textbook QuestionConsider the equilibrium B1aq2 + H2O1l2 Δ HB+1aq2 + OH-1aq2. Suppose that a salt of HB+1aq2 is added to a solution of B1aq2 at equilibrium. (c) Will the pH of the solution increase, decrease, or stay the same?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
338
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos