18. Aqueous Equilibrium
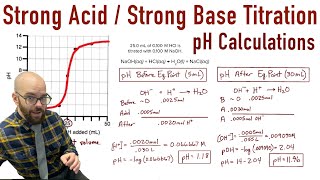
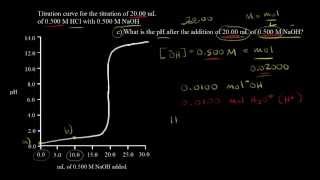
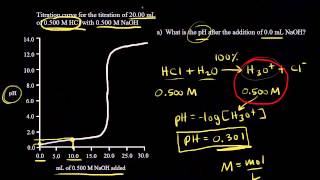
Titrations: Strong Acid-Strong Base

Get help from an AI Tutor
Ask a question to get started.
Problem 118
Textbook Question
Textbook QuestionThe volume of an adult's stomach ranges from about 50 mL when empty to 1 L when full. If the stomach volume is 400 mL and its contents have a pH of 2, how many moles of H+ does the stomach contain? Assuming that all the H+ comes from HCl, how many grams of sodium hydrogen carbonate will totally neutralize the stomach acid?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
704
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos