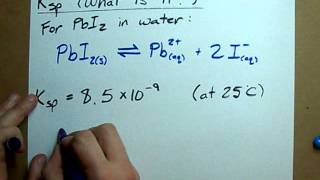18. Aqueous Equilibrium
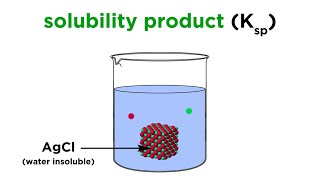
Solubility Product Constant: Ksp

Get help from an AI Tutor
Ask a question to get started.
Problem 111
Textbook Question
Textbook QuestionCalculate the molar solubility of PbCrO4 in: (a) Pure Water (d) 1.0 x 10^-3 M K2CrO4
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
909
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos