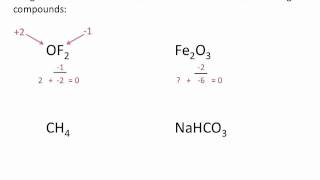6. Chemical Quantities & Aqueous Reactions
Calculate Oxidation Numbers

Get help from an AI Tutor
Ask a question to get started.
Problem 113c
Textbook Question
Textbook QuestionAqueous solutions of ammonia 1NH32 and bleach (active ingredient NaOCl) are sold as cleaning fluids, but bottles of both of them warn: 'Never mix ammonia and bleach, as toxic gases may be produced.' One of the toxic gases that can be produced is chloroamine, NH2Cl. (b) What is the oxidation number of chlorine in chloramine?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
243
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos