11. Bonding & Molecular Structure
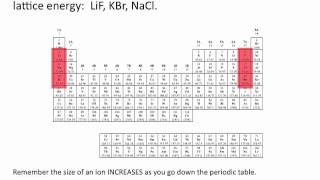
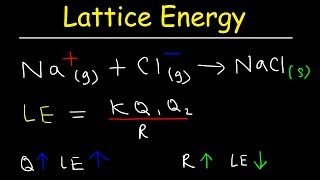
Lattice Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 79
Textbook Question
Textbook QuestionConsider the lattice energies of the following Group 2A compounds: BeH2, 3205 kJ/mol; MgH2, 2791 kJ/mol; CaH2, 2410 kJ/mol; SrH2, 2250 kJ/mol; BaH2, 2121 kJ/mol. (a) What is the oxidation number of H in these compounds?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
658
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos