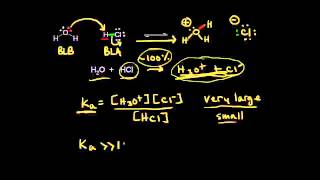
17. Acid and Base Equilibrium
Ka and Kb

Get help from an AI Tutor
Ask a question to get started.
Problem 102
Textbook Question
Textbook QuestionWhich, if any, of the following statements are true? (a) The stronger the base, the smaller the pKb. (b) The stronger the base, the larger the pKb. (c) The stronger the base, the smaller the Kb. (d) The stronger the base, the larger the Kb. (e) The stronger the base, the smaller the pKa of its conjugate acid. (f) The stronger the base, the larger the pKa of its conjugate acid.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1505
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos