6. Chemical Quantities & Aqueous Reactions
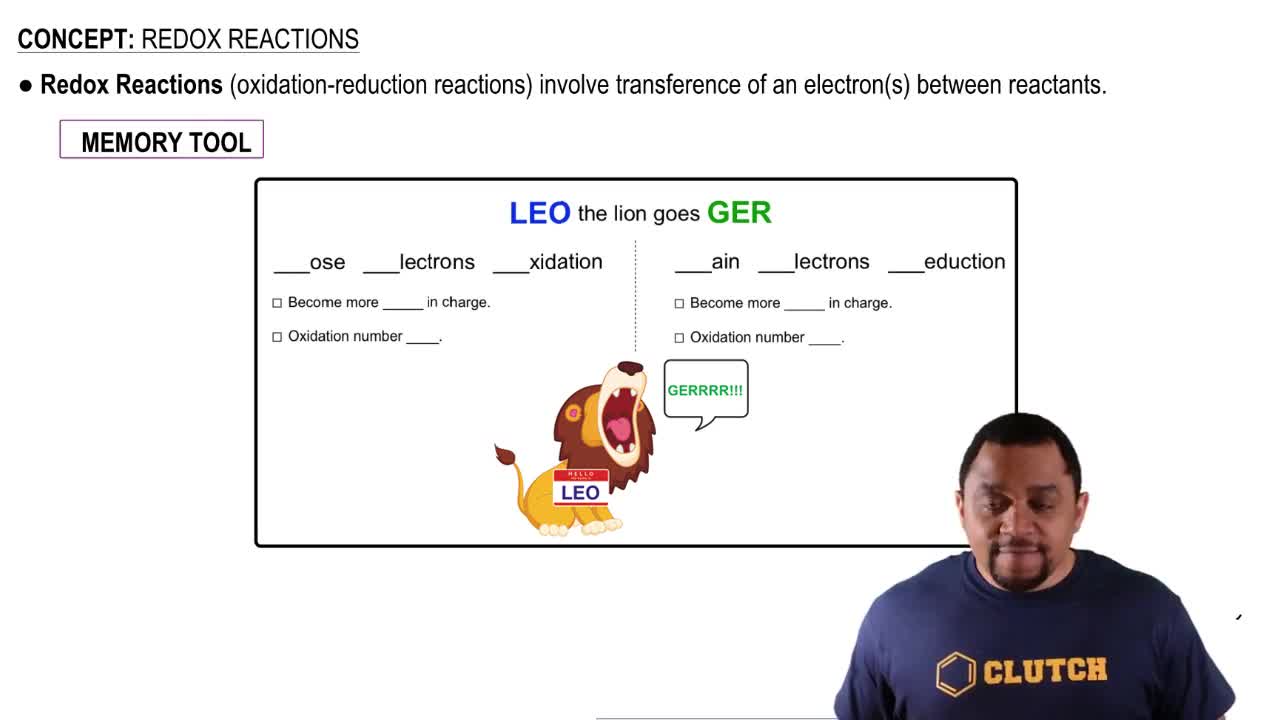
Redox Reactions

Get help from an AI Tutor
Ask a question to get started.
Problem 125a
Textbook Question
Textbook QuestionA sample of impure tin of mass 0.535 g is dissolved in strong acid to give a solution of Sn2+. The solution is then titrated with a 0.0448 M solution of NO3-, which is reduced to NO(g). The equivalence point is reached upon the addition of 0.0344 L of the NO3- solution. Find the percent by mass of tin in the original sample, assuming that it contains no other reducing agents.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
688
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos