8. Thermochemistry
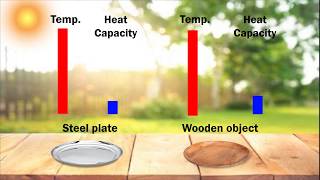
Heat Capacity

Get help from an AI Tutor
Ask a question to get started.
Multiple Choice
Multiple Choice50.00 g of heated metal ore is placed into an insulated beaker containing 822.5 g of water. Once the metal heats up the final temperature of the water is 32.08 ºC. If the metal gains 14.55 kJ of energy, what is the initial temperature of the water? The specific heat capacity of copper is 4.184 J / g ºC.
A
23.86 °C
B
32.86 °C
C
63.08 °C
D
36.31 °C
763
views
8
rank
4
comments
Related Videos
Related Practice
Showing 1 of 14 videos