7. Gases
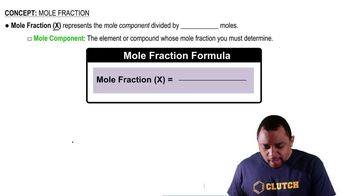
Mole Fraction

Get help from an AI Tutor
Ask a question to get started.
Problem 106
Textbook Question
Textbook QuestionFluorocarbons (compounds that contain both carbon and fluorine) were, until recently, used as refrigerants. The compounds listed in the following table are all gases at 25 °C, and their solubilities in water at 25 °C and 1 atm fluorocarbon pressure are given as mass percentages. (c) Infants born with severe respiratory problems are sometimes given liquid ventilation: They breathe a liquid that can dissolve more oxygen than air can hold. One of these liquids is a fluorinated compound, CF3(CF2)7Br. The solubility of oxygen in this liquid is 66 mL O2 per 100 mL liquid. In contrast, air is 21% oxygen by volume. Calculate the moles of O 2 present in an infant's lungs (volume: 15 mL) if the infant takes a full breath of air compared to taking a full 'breath' of a saturated solution of O 2 in the fluorinated liquid. Assume a pressure of 1 atm in the lungs. Fluorocarbon Solubility (mass %) CF4 0.0015 CClF3 0.009 CCl2F2 0.028 CHClF2 0.30
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
364
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos