8. Thermochemistry
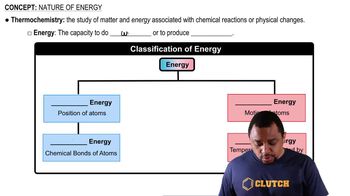
Nature of Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 116
Textbook Question
Textbook QuestionSucrose (C12H22O11) is produced by plants as follows: 12 CO21g2 + 11 H2O1l2¡ C12H22O11 + 12 O21g2 H = 5645 kJ About 4.8 g of sucrose is produced per day per square meter of the earth's surface. The energy for this endothermic reaction is supplied by the sunlight. About 0.1 % of the sunlight that reaches the earth is used to produce sucrose. Calculate the total energy the sun supplies for each square meter of surface area. Give your answer in kilowatts per square meter 1kW>m2 where 1W = 1 J>s2.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
635
views
Was this helpful?
Related Videos
Related Practice