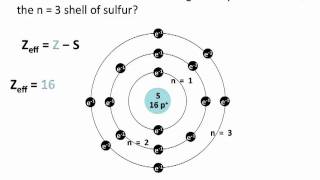
10. Periodic Properties of the Elements
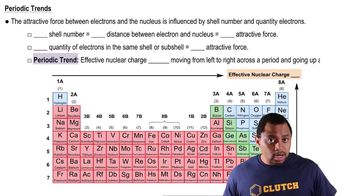
Periodic Trend: Effective Nuclear Charge

Get help from an AI Tutor
Ask a question to get started.
Multiple Choice
Multiple ChoiceUsing Slater's Rules calculate the effective nuclear charge of the 4d orbital electron in iodine.
A
26.1%
B
31.8%
C
70.1%
D
72.7%
E
82.1%
1810
views
2
comments
Related Videos
Related Practice
Showing 1 of 12 videos