10. Periodic Properties of the Elements
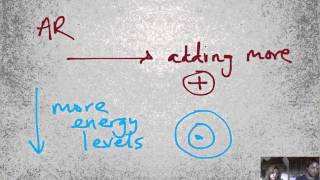
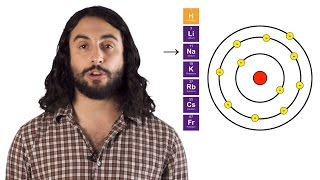
Periodic Trend: Atomic Radius

Get help from an AI Tutor
Ask a question to get started.
Problem 84
Textbook Question
Textbook QuestionIn Table 7.8, the bonding atomic radius of neon is listed as 58 pm, whereas that for xenon is listed as 140 pm. A classmate of yours states that the value for Xe is more realistic than the one for Ne. Is she correct? If so, what is the basis for her statement?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1014
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos