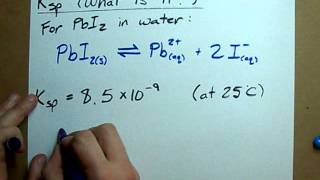18. Aqueous Equilibrium
Solubility Product Constant: Ksp

Get help from an AI Tutor
Ask a question to get started.
Problem 10
Textbook Question
Textbook QuestionCa1OH22 has a Ksp of 6.5 * 10-6. (b) If 50 mL of the solution from part (a) is added to each of the beakers shown here, in which beakers, if any, will a precipitate form? In those cases where a precipitate forms, what is its identity? [Section 17.6]

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
17mPlay a video:
571
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos