2. Atoms & Elements
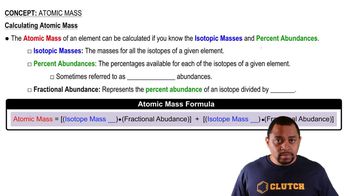
Atomic Mass

Get help from an AI Tutor
Ask a question to get started.
Problem 120b
Textbook Question
Textbook QuestionNaturally occurring chlorine is composed of two isotopes: 75.76% Cl-35 (mass 34.9688 amu) and 24.24% Cl-37 (mass 36.9659 amu). Naturally occurring oxygen is composed of three isotopes: 99.757% O-16 (mass 15.9949 amu), 0.038% O-17 (mass 16.9991 amu), and 0.205% O-18 (mass 17.9991 amu). The compound dichlorine monoxide is composed of two chlorine atoms and one oxygen atom bonded together to form the Cl2O molecule. How many Cl2O molecules of different masses naturally exist? Give the masses of the three most abundant Cl2O molecules.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1678
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos