2. Atoms & Elements
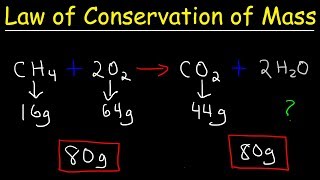
Law of Conservation of Mass

Get help from an AI Tutor
Ask a question to get started.
Problem 80
Textbook Question
Textbook QuestionOne of the steps in the commercial process for converting ammonia to nitric acid is the conversion of NH3 to NO: 4 NH31g2 + 5 O21g2¡4 NO1g2 + 6 H2O1g2 In a certain experiment, 2.00 g of NH3 reacts with 2.50 g of O2. (d) Show that your calculations in parts (b) and (c) are consistent with the law of conservation of mass.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1696
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos