12. Molecular Shapes & Valence Bond Theory
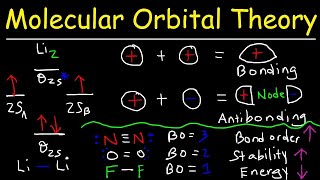
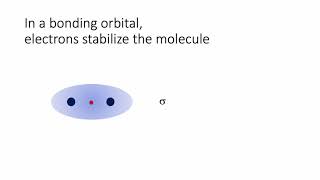
Molecular Orbital Theory

Get help from an AI Tutor
Ask a question to get started.
Problem 71
Textbook Question
Textbook QuestionConsider the H2+ ion. (f) Which of the following statements about part (e) is correct: (i) The light excites an electron from a bonding orbital to an antibonding orbital, (ii) The light excites an electron from an antibonding orbital to a bonding orbital, or (iii) In the excited state there are more bonding electrons than antibonding electrons?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
442
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos