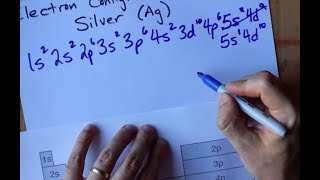10. Periodic Properties of the Elements
The Electron Configuration: Condensed

Get help from an AI Tutor
Ask a question to get started.
Problem 79
Textbook Question
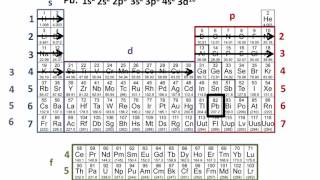
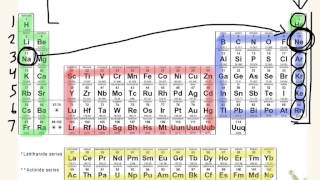
Textbook QuestionThe following do not represent valid ground-state electron configurations for an atom either because they violate the Pauli exclusion principle or because orbitals are not filled in order of increasing energy. Indicate which of these two principles is violated in each example. (b) 3Xe46s3
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
580
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos