2. Atoms & Elements
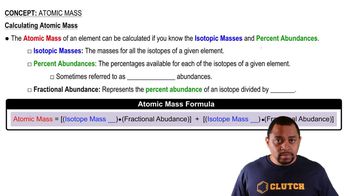
Atomic Mass

Get help from an AI Tutor
Ask a question to get started.
Problem 96
Textbook Question
Textbook QuestionCopper (Cu) consists of two naturally occurring isotopes with masses of 62.9296 and 64.9278 u. (a) How many protons and neutrons are in the nucleus of each isotope? Write the complete atomic symbol for each, showing the atomic number and mass number. (b) The average atomic mass of Cu is 63.55 u. Calculate the abundance of each isotope.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1638
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos