9. Quantum Mechanics
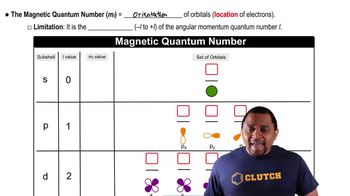
Quantum Numbers: Magnetic Quantum Number

Get help from an AI Tutor
Ask a question to get started.
Multiple Choice
Multiple ChoiceWhich of the following statements is false?
a) A set of d orbitals contains 5 orbitals.
b) A set of 4s orbitals would have more energy than a set of 3p orbitals.
c) The second shell of an atom possesses d orbitals.
d) A set of f orbitals contains 3 orbitals.
e) The first energy level contains only s orbitals.
A
a, b
B
b, d
C
c, d
D
d, e
E
b, e
566
views
4
rank
1
comments
Related Videos
Related Practice
Showing 1 of 10 videos