8. Thermochemistry
Hess's Law

Get help from an AI Tutor
Ask a question to get started.
Problem 62b
Textbook Question
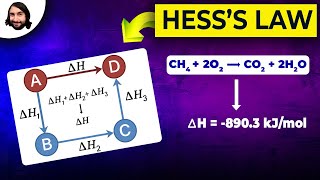
Textbook QuestionConsider the following hypothetical reactions: A ¡B HI = +60 kJ B ¡ C HII = -90 kJ (b) Construct an enthalpy diagram for substances A, B, and C, and show how Hess's law applies.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
1052
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos