8. Thermochemistry
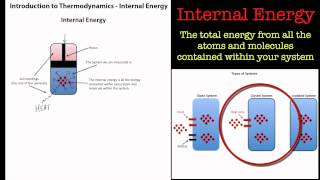
Internal Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 39a
Textbook Question
Textbook QuestionIdentify each energy exchange as primarily heat or work and determine whether the sign of ΔE is positive or negative for the system. a. Sweat evaporates from skin, cooling the skin. (The evaporating sweat is the system.) b. A balloon expands against an external pressure. (The contents of the balloon is the system.) c. An aqueous chemical reaction mixture is warmed with an external flame. (The reaction mixture is the system.) Identify energy exchanges as primarily heat or work. Determine whether the sign of E is positive or negative for the system.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1055
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos