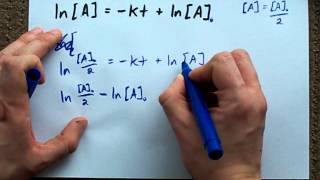21. Nuclear Chemistry
Rate of Radioactive Decay

Get help from an AI Tutor
Ask a question to get started.
Problem 52
Textbook Question
Textbook QuestionBased on the following atomic mass values:1H, 1.00782 amu; 2H, 2.01410 amu; 3H, 3.01605 amu; 3He, 3.01603 amu; 4He, 4.00260 amu—and the mass of the neutron given in the text, calculate the energy released per mole in each of the following nuclear reactions, all of which are possibilities for a controlled fusion process: (b) 21H + 21H¡32He + 10n
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
520
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos