19. Chemical Thermodynamics
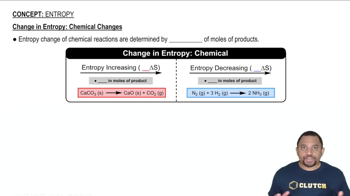
Entropy

Get help from an AI Tutor
Ask a question to get started.
Problem 78
Textbook Question
Textbook QuestionFor the vaporization of benzene, ∆Hvap = 30.7 kJ/mol and ∆Svap = 87.0 J/(K*mol). Calculate ∆Ssurr and ∆Stotal at: (a) 70 °C
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
512
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos