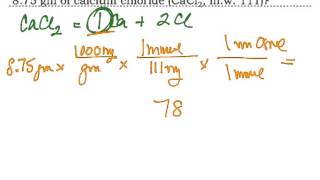6. Chemical Quantities & Aqueous Reactions
Osmolarity

Get help from an AI Tutor
Ask a question to get started.
Problem 70
Textbook Question
Textbook QuestionRinger's solution, used in the treatment of burns and wounds, is prepared by dissolving 4.30 g of NaCl, 0.150 g of KCl, and 0.165 g of CaCl2 in water and diluting to a volume of 500.0 mL. What is the molarity of each of the component ions in the solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
797
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos