19. Chemical Thermodynamics
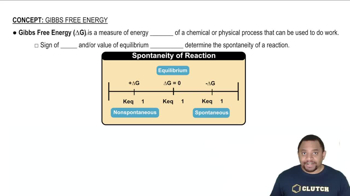
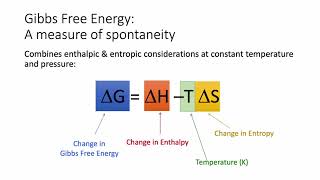
Gibbs Free Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 15a
Textbook Question
Textbook QuestionAmmonium hydrogen sulfide, a stink bomb ingredient, decomposes to ammonia and hydrogen sulfide: Calculate the standard free-energy change for the rection at 25 °C if the total pressure resulting from the solid NH4S placed in an evacuated container is 0.658 atm at 25 °C. (a) -43.8 kJ (b) +1.04 kJ (c) -462 kJ (d) +5.51 kJ
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
311
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos