2. Atoms & Elements
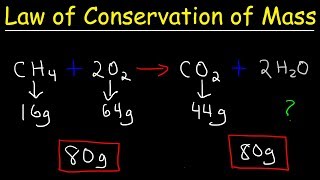
Law of Conservation of Mass

Get help from an AI Tutor
Ask a question to get started.
Problem 79
Textbook Question
Textbook QuestionA sample of CaCO3 was heated, causing it to form CaO and CO2 gas. Solid CaO remained behind, while the CO2 escaped to the atmosphere. If the CaCO3 weighed 612 g and the CaO weighed 343 g, how many grams of CO2 were formed in the reaction?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
350
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos