7. Gases
Partial Pressure

Get help from an AI Tutor
Ask a question to get started.
Problem 108
Textbook Question
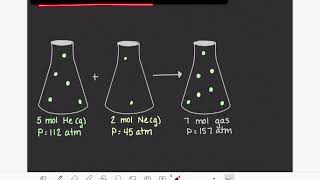
Textbook QuestionAn 8.40-g sample of argon and an unknown mass of H2 are mixed in a flask at room temperature. The partial pressure of the argon is 44.0 kPa, and that of the hydrogen is 57.33 kPa. What is the mass of the hydrogen?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
452
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos