19. Chemical Thermodynamics
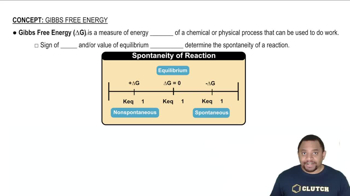
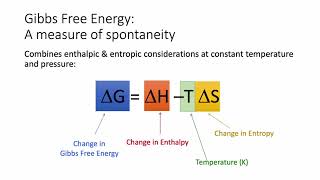
Gibbs Free Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 108a
Textbook Question
Textbook QuestionUse the data in Appendix B to calculate ∆G for the decom-position of nitrosyl chloride at 25 °C when the partial pressures are 2.00 atm of NOCl, 1.00 * 10^-3 atm of NO, and 1.00 * 10^-3 atm of Cl2: Is the reaction spontaneous in the forward or the reverse direction under these conditions?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
158
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos