8. Thermochemistry
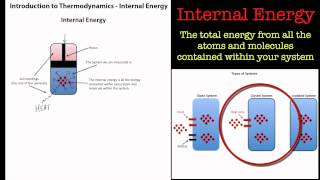
Internal Energy

Get help from an AI Tutor
Ask a question to get started.
Problem 44
Textbook Question
Textbook QuestionThe air in an inflated balloon (defined as the system) warms over a toaster and absorbs 115 J of heat. As it expands, it does 77 kJ of work. What is the change in internal energy for the system?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1535
views
2
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos