7. Gases
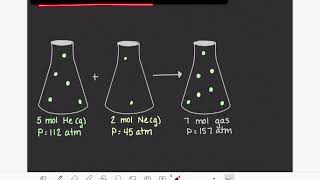
Partial Pressure

Get help from an AI Tutor
Ask a question to get started.
Problem 81
Textbook Question
Textbook QuestionNatural gas is a mixture of many substances, primarily CH4, C2H6, C3Hg, and C4H10. Assuming that the total pressure of the gases is 1.48 atm and that their mole ratio is 94:4.0:1.5:0.50, calculate the partial pressure in atmospheres of each gas.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
571
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos