6. Chemical Quantities & Aqueous Reactions
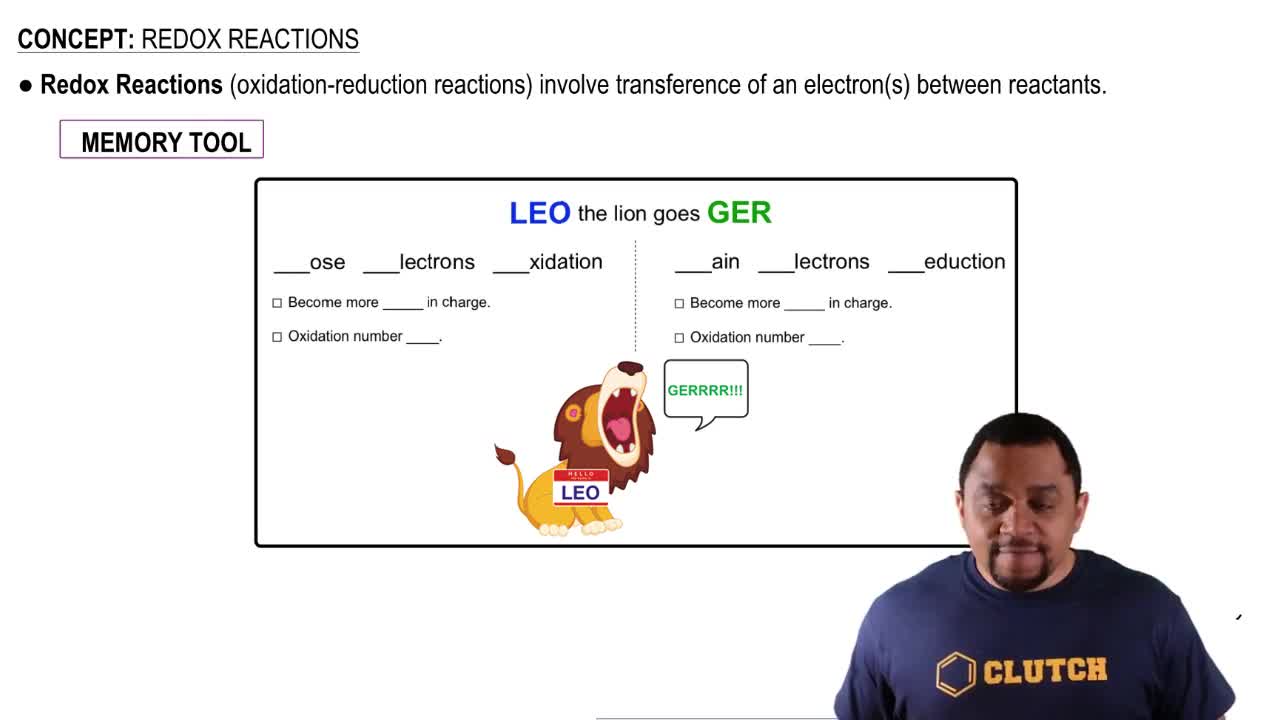
Redox Reactions

Get help from an AI Tutor
Ask a question to get started.
Problem 163
Textbook Question
Textbook QuestionGold metal is extracted from its ore by treating the crushed rock with an aerated cyanide solution. The unbalanced equation for the reaction is (b) Use any of the following data at 25 °C to calculate ∆G° for this reaction at 25 °C: Kf for Au(CN)2- = 6.2 x 10^38, Ka for HCN = 4.9 x 10^-10, and standard reduction potentials are
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
11mPlay a video:
389
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos