11. Bonding & Molecular Structure
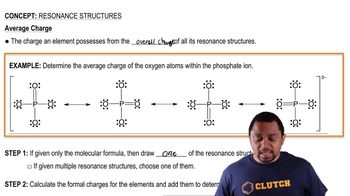
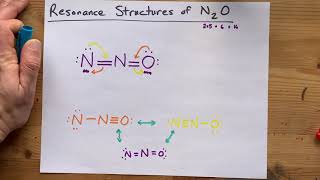
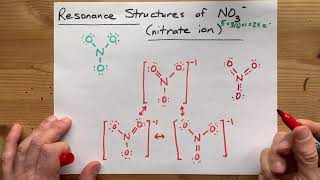
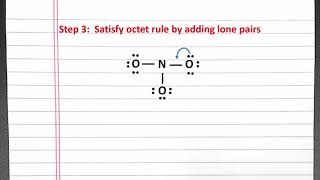
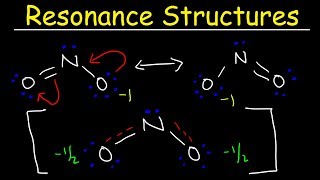
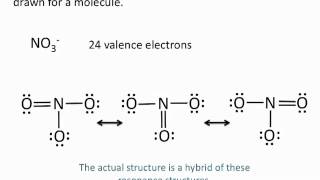
Resonance Structures

Get help from an AI Tutor
Ask a question to get started.
Problem 111
Textbook Question
Textbook QuestionIn the cyanate ion, OCN-, carbon is the central atom. (b) Which resonance structure makes the greatest contribution to the resonance hybrid? Which makes the least contribution? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
1026
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos