13. Liquids, Solids & Intermolecular Forces
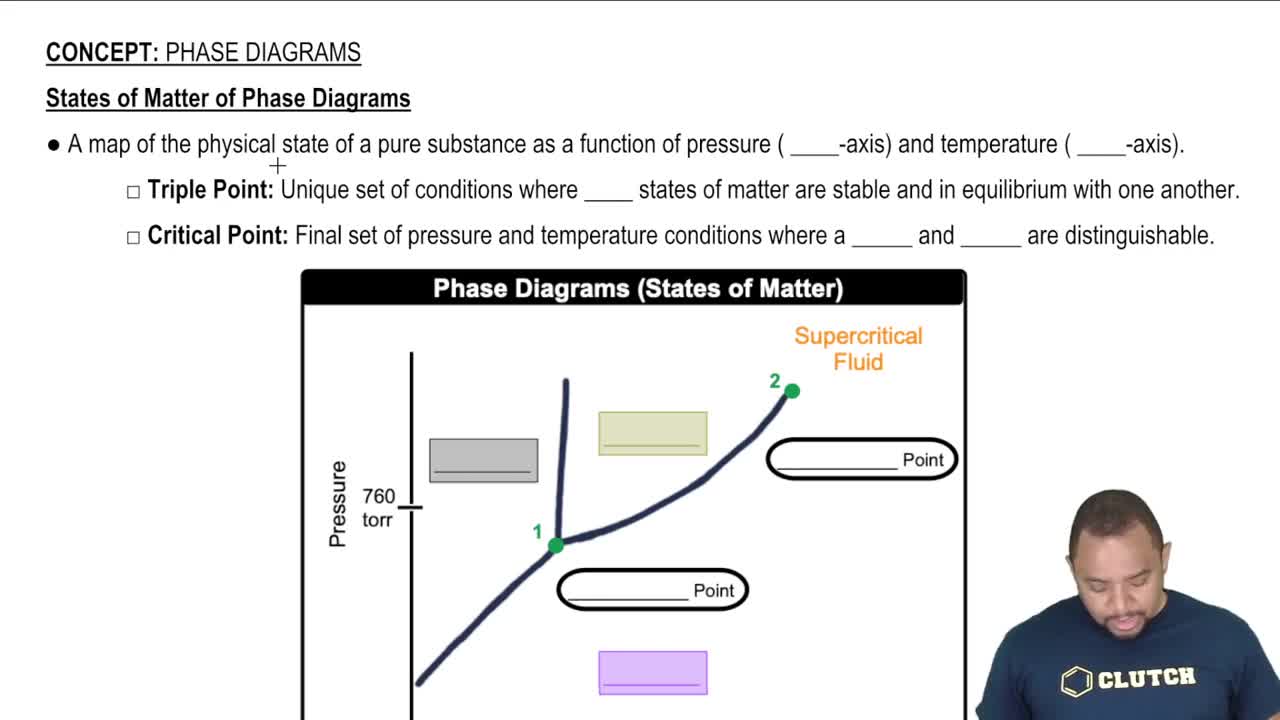
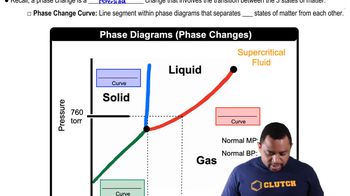
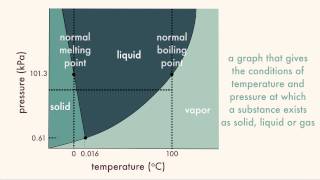
Phase Diagrams

Get help from an AI Tutor
Ask a question to get started.
Problem 14
Textbook Question
Textbook QuestionBenzoic acid, C6H5COOH, melts at 122 °C. The density in the liquid state at 130 °C is 1.08 g/cm3. The density of solid benzoic acid at 15 °C is 1.266 g/cm3. (b) If you converted a cubic centimeter of liquid benzoic acid into a solid, would the solid take up more, or less, volume than the original cubic centimeter of liquid?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1270
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos