7. Gases
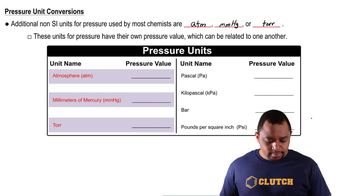
Pressure Units

Get help from an AI Tutor
Ask a question to get started.
Problem 97
Textbook Question
Textbook QuestionTorricelli, who invented the barometer, used mercury in its construction because mercury has a very high density, which makes it possible to make a more compact barometer than one based on a less dense fluid. Calculate the density of mercury using the observation that the column of mercury is 760 mm high when the atmospheric pressure is 1.01 * 105 Pa. Assume the tube containing the mercury is a cylinder with a constant cross-sectional area.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
571
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos