19. Chemical Thermodynamics
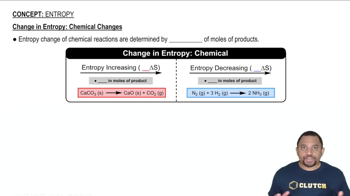
Entropy

Get help from an AI Tutor
Ask a question to get started.
Problem 26d
Textbook Question
Textbook Question(b) In a particular spontaneous process the entropy of the system decreases. What can you conclude about the sign and magnitude of ΔSsurr?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
56sPlay a video:
240
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos