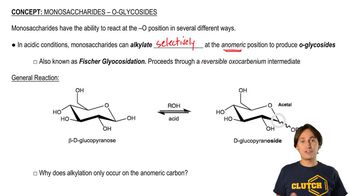
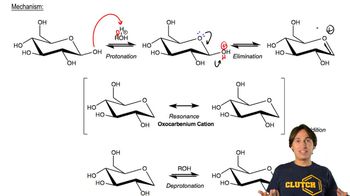
24. Carbohydrates
Glycoside
Problem 23b
Textbook Question
Textbook QuestionSome protecting groups can block two OH groups of a carbohydrate at the same time. One such group is shown here, protecting the 4-OH and 6-OH groups of b-d-glucose.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
289
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos