13. Liquids, Solids & Intermolecular Forces
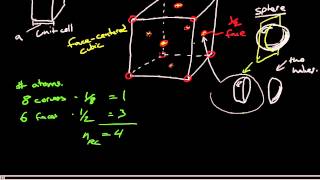
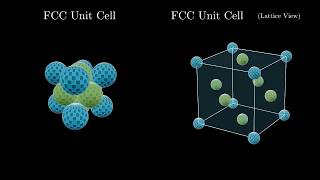
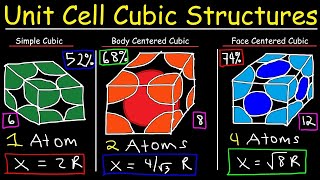
Face Centered Cubic Unit Cell

Get help from an AI Tutor
Ask a question to get started.
Problem 36
Textbook Question
Textbook QuestionCopper crystallizes in a face-centered cubic unit cell with an edge length of 362 pm. What is the radius of a copper atom in picometers? What is the density of copper in g>cm3?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
829
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos