 Back
BackAcid-Base Balance and Arterial Blood Gas Analysis
Study Guide - Smart Notes

Acid-Base Balance
Introduction to Acid-Base Balance
The acid-base balance refers to the regulation of hydrogen ion concentration (H+) in the blood, which is measured by the pH. The normal physiological range for blood pH is 7.35 (venous) to 7.45 (arterial). Maintaining this range is crucial for proper cellular function, and deviations can compromise organ systems. The body uses several mechanisms to keep blood pH constant.
Key Systems in Acid-Base Regulation
Respiratory System: The lungs regulate blood pH by controlling the amount of carbon dioxide (CO2) exhaled. Chemoreceptors in the medulla oblongata detect CO2 levels and adjust respiration rate and depth accordingly.
Renal System: The kidneys manage blood pH by excreting or reabsorbing hydrogen ions and bicarbonate (HCO3-). Metabolic processes produce CO2, H2O, and H+, which are buffered and excreted as needed.
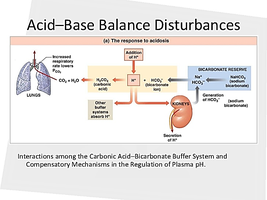
The Carbonic Acid-Bicarbonate Buffer System
The primary chemical equation governing acid-base balance is:
The left side (CO2 + H2O) is influenced by the respiratory system.
The right side (HCO3- + H+) is influenced by the renal system.
Changes in respiration or kidney function shift the equation, affecting blood pH.
Arterial Blood Gas (ABG) Analysis
Purpose and Units
ABG analysis is used to assess oxygenation status and acid-base balance in clinical settings. Gas values are measured in mm Hg, while bicarbonate is reported in meq/L or mmol/L (equivalent units).
Normal Adult Arterial Values
Base Excess (BE): Indicates the amount of H+ needed to return pH to 7.35 if pCO2 is normal.
HCO3-: Bicarbonate concentration.
PO2: Partial pressure of oxygen.
PCO2: Partial pressure of carbon dioxide.
SO2: Oxygen saturation.

Six Steps to ABG Analysis
Is the pH out of range?
Is the pCO2 normal?
Is the HCO3- out of range?
Match the abnormal result with the pH.
Does the pCO2 or HCO3- go in the opposite or same direction as pH?
Is the pO2 and O2 saturation out of range?
Compensation Mechanisms
Respiratory Compensation: The lungs adjust CO2 levels to correct pH.
Metabolic Compensation: The kidneys adjust HCO3- levels to correct pH.
If CO2 or HCO3- moves opposite to pH, compensation is occurring.
Clinical Conditions Affecting Acid-Base Balance
Respiratory Alkalosis
Occurs with excessive breathing (hyperventilation), leading to decreased CO2 and increased pH (alkalosis).
Causes: Voluntary hyperventilation, stimulation of respiratory center, artificial ventilation.
Mechanism: CO2 is blown off, equation shifts left, H+ decreases, pH rises.
Respiratory Acidosis
Occurs with poor lung perfusion, impaired gas exchange, or decreased ventilation, leading to increased CO2 and decreased pH (acidosis).
Mechanism: Less CO2 is exhaled, equation shifts right, H+ increases, pH falls.
Metabolic Alkalosis
Occurs with loss of H+ (e.g., vomiting) or ingestion of bicarbonate, leading to increased pH.
Compensation: Lungs decrease respiration to retain CO2 and restore pH.
Metabolic Acidosis
Occurs with increased H+ production (e.g., diabetic ketoacidosis), decreased H+ loss (renal failure), or loss of bicarbonate (vomiting), leading to decreased pH.
Compensation: Lungs increase respiration to blow off CO2 and restore pH.
Clinical Examples
Example 1: Respiratory Alkalosis
Mr. Jones, after a panic attack, has a respiratory rate of 45. ABG results: pH 7.55, pCO2 16.72 mmHg, pO2 114.76 mmHg.
Interpretation: High pH, low pCO2 indicates respiratory alkalosis.
Example 2: Respiratory Acidosis
Mrs. Parker, with suspected pulmonary embolism, ABG results: pH 7.21, pCO2 74.4 mmHg, pO2 58.52 mmHg, bicarbonate 28 mmol/L, base excess -1.
Interpretation: Low pH, high pCO2 indicates respiratory acidosis.
Summary
The lungs and kidneys collaborate to maintain blood pH. When one system is compromised, the other compensates. ABG analysis is essential for evaluating acid-base status in clinical practice. Respiratory compensation is immediate, while metabolic compensation may take up to two days.
Condition | Primary Change | Compensation |
|---|---|---|
Respiratory Alkalosis | ↓ CO2, ↑ pH | Kidneys excrete HCO3- |
Respiratory Acidosis | ↑ CO2, ↓ pH | Kidneys retain HCO3- |
Metabolic Alkalosis | ↑ HCO3-, ↑ pH | Lungs decrease ventilation |
Metabolic Acidosis | ↓ HCO3-, ↓ pH | Lungs increase ventilation |
Additional info: The detailed mechanisms of bicarbonate and hydrogen regulation in the kidney are complex and can be found in medical textbooks. For basic understanding, focus on the kidneys' role in reabsorbing bicarbonate and excreting hydrogen ions.
