 Back
BackAdrenal Cortex: Structure, Function, and Clinical Relevance
Study Guide - Smart Notes

Adrenal Gland Structure and Morphology
Gross and Microscopic Anatomy
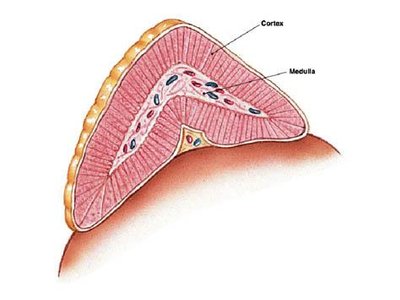
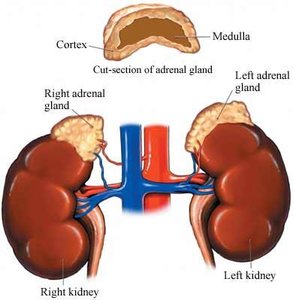
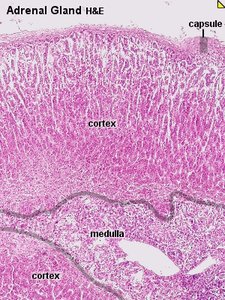
The adrenal glands are paired endocrine organs located superior to the kidneys in mammals. Each gland consists of two distinct regions: the outer adrenal cortex and the inner adrenal medulla. The cortex is responsible for steroid hormone production, while the medulla produces catecholamines.
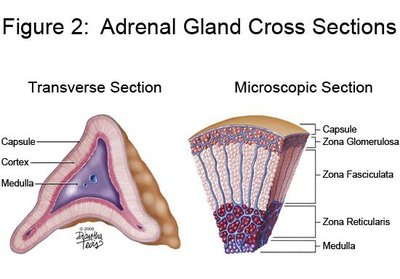
Adrenal Cortex: The outer layer, subdivided into three zones: zona glomerulosa, zona fasciculata, and zona reticularis.
Adrenal Medulla: The inner region, functionally and embryologically distinct from the cortex.
Functional Zonation of the Cortex
The adrenal cortex is divided into three zones, each responsible for synthesizing specific steroid hormones:
Zona Glomerulosa: Produces mineralocorticoids (e.g., aldosterone).
Zona Fasciculata: Produces glucocorticoids (e.g., cortisol).
Zona Reticularis: Produces androgens (e.g., DHEA).
Adrenal Cortex Steroidogenesis
Overview of Steroid Hormones
The adrenal cortex synthesizes three main classes of steroid hormones from cholesterol:
Glucocorticoids: Regulate metabolism and immune response (e.g., cortisol).
Mineralocorticoids: Regulate sodium and potassium balance (e.g., aldosterone).
Androgens: Weak sex steroids (e.g., DHEA).
Cholesterol as the Precursor
Cholesterol is the substrate for all steroid hormone biosynthesis. It is transported into mitochondria, where the side-chain cleavage enzyme (CYP11A1) converts it to pregnenolone, the first steroid in the pathway. 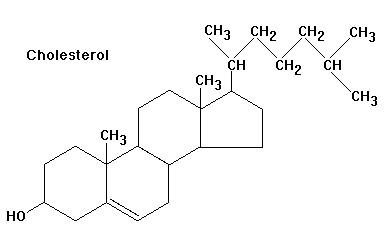
Steroid Biosynthesis Pathways
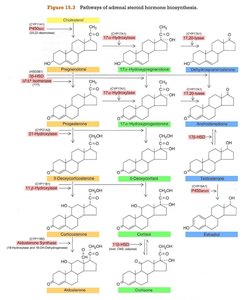
The conversion of cholesterol to various steroid hormones involves several enzymes, including hydroxylases and dehydrogenases. The specific pathway depends on the enzymes present in each cortical zone.
Key Enzymes: CYP11A1 (side-chain cleavage), 3β-HSD, CYP17, CYP21A2, CYP11B1, CYP11B2, CYP19 (aromatase).
Pathway Branching: Zona glomerulosa lacks CYP17, so it cannot produce glucocorticoids or androgens.
Cellular Features of Steroidogenic Cells
Steroidogenic cells in the adrenal cortex are characterized by abundant mitochondria and smooth endoplasmic reticulum (SER), reflecting their role in steroid biosynthesis.
Lipid Droplets: Store cholesterol esters for hormone production.
Mitochondria: Site of initial cholesterol conversion to pregnenolone.
SER: Site of further steroid modifications.
Regulation of Adrenal Cortex Function
Zona-Specific Regulation
Zona Glomerulosa: Regulated primarily by the renin-angiotensin-aldosterone system (RAAS) and plasma potassium levels.
Zona Fasciculata and Reticularis: Regulated by adrenocorticotropic hormone (ACTH) from the anterior pituitary.
Hypothalamic-Pituitary-Adrenal (HPA) Axis
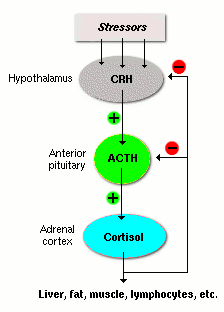
The HPA axis controls glucocorticoid secretion via a negative feedback loop:
Stress or circadian signals stimulate the hypothalamus to release corticotropin-releasing hormone (CRH).
CRH stimulates the anterior pituitary to secrete ACTH.
ACTH stimulates the adrenal cortex to produce cortisol.
Cortisol exerts negative feedback on both the hypothalamus and pituitary.
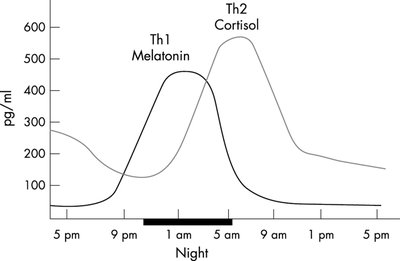
Circadian and Stress-Induced Secretion
Circadian Rhythm: ACTH and cortisol are secreted in a daily rhythm, peaking in the early morning.
Stress Response: ACTH and cortisol levels rise rapidly in response to stressors.
Physiological Actions of Adrenal Steroids
Glucocorticoids (Cortisol)
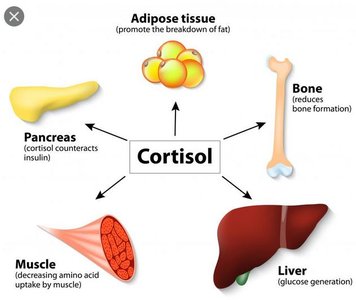
Cortisol is the primary glucocorticoid in humans, affecting metabolism, immune function, and stress adaptation.
Metabolic Effects: Stimulates gluconeogenesis in the liver, increases blood glucose, promotes lipolysis, and induces protein catabolism in muscle.
Immune Effects: Suppresses inflammation and immune responses at pharmacological levels.
Permissive Actions: Enhances the effects of catecholamines and other hormones during stress.
Mineralocorticoids (Aldosterone)
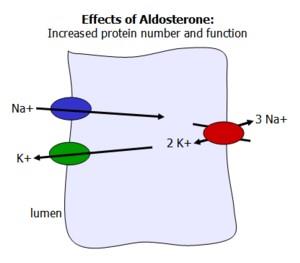
Aldosterone acts on the distal tubules of the kidney to increase sodium reabsorption and potassium excretion, thereby regulating blood pressure and fluid balance.
Mechanism: Increases the number and activity of sodium-potassium pumps in renal epithelial cells.
Androgens
The adrenal cortex produces weak androgens (e.g., DHEA, DHEA-S), which are precursors for sex steroids and have minor effects compared to gonadal androgens.
Developmental Aspects of the Adrenal Cortex
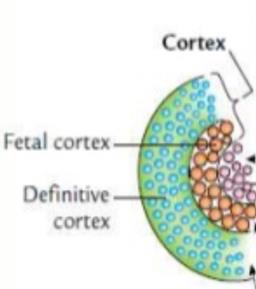
Fetal Adrenal Gland
Fetal adrenal glands are proportionally larger than adult glands and consist of a large fetal zone and a smaller permanent cortex.
The fetal zone produces DHEA-S, a precursor for placental estrogen synthesis.
After birth, the fetal zone regresses, and the adult cortical zones differentiate.
Pathophysiology of Adrenocortical Function
Adrenocortical Hypofunction (Addison's Disease)
Primary: Destruction of the adrenal cortex leads to deficiencies in glucocorticoids, mineralocorticoids, and androgens.
Symptoms: Weakness, weight loss, hypotension, hyperpigmentation, salt craving, hypoglycemia.

Adrenocortical Hyperfunction (Cushing's Syndrome and Conn's Syndrome)
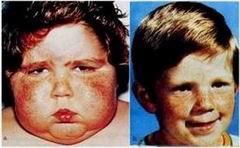
Cushing's Syndrome: Excess cortisol production due to adrenal or pituitary tumors. Symptoms include centripetal obesity, muscle weakness, hypertension, and skin changes.
Conn's Syndrome: Excess aldosterone production, usually from an adrenal adenoma, leading to hypertension, hypokalemia, and metabolic alkalosis.
Summary Table: Adrenal Cortex Zones and Hormones
Zone | Main Hormone(s) | Main Function |
|---|---|---|
Zona Glomerulosa | Aldosterone (mineralocorticoid) | Regulates Na+/K+ balance, blood pressure |
Zona Fasciculata | Cortisol (glucocorticoid) | Regulates metabolism, stress response |
Zona Reticularis | DHEA, DHEA-S (androgens) | Precursor for sex steroids |
Key Equations
Glucocorticoid Negative Feedback: $ \text{CRH} \xrightarrow{+} \text{ACTH} \xrightarrow{+} \text{Cortisol} \xrightarrow{-} \text{CRH, ACTH} $
Renin-Angiotensin-Aldosterone System: $ \text{Angiotensinogen} \xrightarrow{Renin} \text{Angiotensin I} \xrightarrow{ACE} \text{Angiotensin II} \xrightarrow{+} \text{Aldosterone} $
Additional info:
Permissive actions of glucocorticoids are essential for the full physiological response to catecholamines during stress.
Chronic glucocorticoid excess can lead to metabolic syndrome, osteoporosis, and immune suppression.
