 Back
BackCell Membrane Transport: Principles and Mechanisms
Study Guide - Smart Notes

Cell Membrane Transport
Overview of Membrane Transport
Cell membrane transport is essential for maintaining cellular homeostasis and enabling communication between the intracellular fluid (ICF) and extracellular fluid (ECF). The plasma membrane selectively allows certain molecules to cross, either by simple diffusion or with the assistance of proteins.
ICF and ECF compositions differ: The internal and external environments of the cell have distinct solute concentrations.
Simple diffusion: Some molecules move across the membrane without assistance.
Protein-mediated transport: Other molecules require membrane proteins to facilitate their movement.
Driving Forces for Transport
The movement of molecules across membranes is governed by various driving forces, which determine the direction and rate of transport.
Chemical driving force: Caused by differences in solute concentration across the membrane.
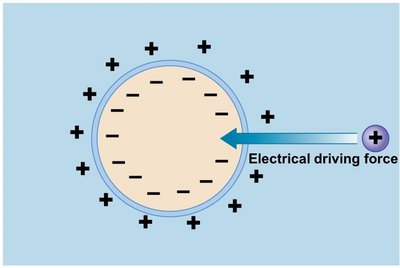
Electrical driving force: Results from differences in charge across the membrane, affecting ions.
Electrochemical driving force: The combined effect of chemical and electrical gradients.


Membrane Potential
Membrane potential (Vm) is the difference in electrical potential across the plasma membrane, determined by the net charge inside the cell. It is crucial for the function of excitable cells such as neurons and muscle cells.
Intracellular fluid: Typically has excess anions (negative charge).
Extracellular fluid: Typically has excess cations (positive charge).

Equilibrium Potential and Electrochemical Gradients
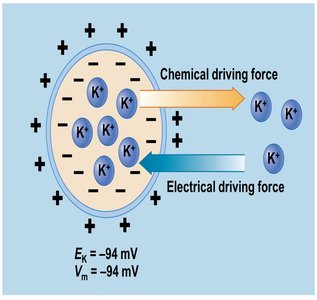
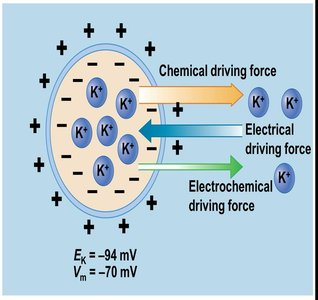
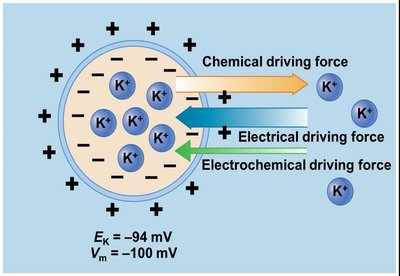
Each ion has an equilibrium potential (EX), the membrane potential at which the electrical and chemical driving forces are balanced, resulting in no net movement of the ion.
Electrochemical gradient: The net driving force for ion movement.
Equilibrium potential examples: ENa = +60 mV, EK = -94 mV.
Net Flux and Types of Transport
Net flux refers to the overall movement of molecules across the membrane, determined by the electrochemical gradient. Transport can be passive or active.
Passive transport: Movement down the electrochemical gradient (simple diffusion, facilitated diffusion, diffusion through channels).
Active transport: Movement up the electrochemical gradient, requiring energy (primary and secondary active transport).

Passive Transport Mechanisms
Simple Diffusion
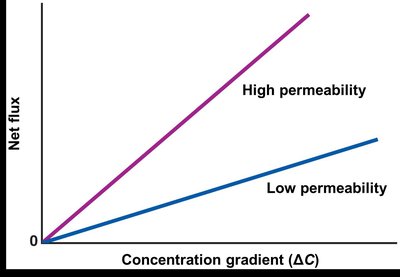
Simple diffusion is the spontaneous movement of molecules across the plasma membrane, driven by the magnitude of the driving force, membrane surface area, and membrane permeability.
Factors affecting rate: Driving force, surface area, permeability.
Diffusional equilibrium: Achieved when concentrations are equal on both sides.


Facilitated Diffusion
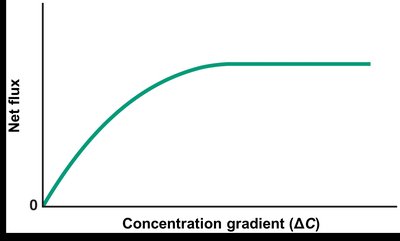
Facilitated diffusion requires transmembrane proteins to transport molecules across the membrane. The rate depends on the transport rate of individual carriers, the concentration gradient, and the number of carriers.
Saturation: When all carriers are occupied, increasing the concentration gradient does not increase transport rate.

Diffusion Through Channels
Channel proteins allow specific molecules, such as ions and water, to pass through the membrane. Channels can be regulated to open or close.
Examples: Aquaporins (water channels), ion channels.

Active Transport Mechanisms
Primary Active Transport
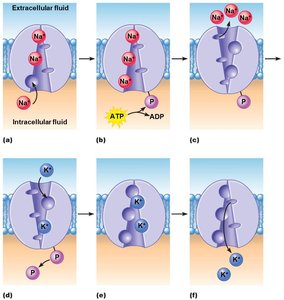
Primary active transport uses ATP directly to move substances against their electrochemical gradient. Pumps act as both carriers and enzymes (ATPase).
Example: Na+/K+ pump.
Mechanism: Pump is phosphorylated and dephosphorylated, causing conformational changes.

Secondary Active Transport
Secondary active transport is powered by an electrochemical gradient created by primary active transport. It includes cotransport (symport) and countertransport (antiport).
Cotransport: Both molecules move in the same direction (e.g., sodium-linked glucose transport).
Countertransport: Molecules move in opposite directions (e.g., sodium-proton exchange).


Osmosis and Water Transport
Osmosis
Osmosis is the passive movement of water across a membrane, driven by differences in solute concentration. It is unaffected by membrane potential and has significant physiological effects on cell volume.
Osmotic driving force: Water moves toward higher solute concentration.
Osmolarity: Total solute particle concentration of a solution.


Osmolarity and Tonicity
Osmolarity refers to the total concentration of solute particles, while tonicity describes the effect of a solution on cell volume, based on the concentration of impermeant solutes.
Iso-osmotic: Equal osmolarity between two solutions.
Hyperosmotic: Higher osmolarity than another solution.
Hypo-osmotic: Lower osmolarity than another solution.
Isotonic: No change in cell volume.
Hypertonic: Cell shrinks.
Hypotonic: Cell swells.
Terms | Definitions and Solute Concentrations |
|---|---|
Iso-osmotic | Total concentration of permeant and impermeant solutes: 300 mOsm |
Hypo-osmotic | Less than 300 mOsm |
Hyperosmotic | Greater than 300 mOsm |
Isotonic | Concentration of impermeant solutes: 300 mOsm |
Hypotonic | Less than 300 mOsm |
Hypertonic | Greater than 300 mOsm |

Clinical Correlates
Excessive Sweating
Excessive sweating increases plasma osmolarity, causing cells to shrink due to water loss.
Water Intoxication
Hyper-hydration decreases plasma osmolarity, leading to cell swelling.
Diabetes Mellitus
In diabetes, impaired insulin-dependent glucose carriers make glucose impermeant, increasing plasma tonicity and causing cell shrinkage and damage.
Vesicular and Epithelial Transport
Vesicular Transport
Vesicular transport involves the movement of substances in membrane-bound compartments, including endocytosis (phagocytosis, pinocytosis, receptor-mediated) and exocytosis.
Endocytosis: Uptake of molecules from ECF into the cell.
Exocytosis: Release of molecules from the cell into the ECF.
Epithelial Transport
Epithelial transport refers to the movement of substances across two plasma membranes, facilitating absorption and secretion between internal and external environments.
Na+-linked co-transport: Glucose is absorbed at the apical membrane via sodium co-transport.
Epithelial water transport: Active solute pumping followed by passive water movement.
Transcytosis
Transcytosis is the transport of macromolecules across epithelial cells, sometimes used as a 'Trojan horse' method for drug delivery.
Areas of Difficulty
Electrochemical gradients
Osmolarity and tonicity
Secondary active transport
Additional info: This guide expands on brief points with academic context, definitions, and examples to ensure completeness and clarity for exam preparation.
