 Back
BackChapter 2: Cell Chemistry & Cell Components – Key Study Notes
Study Guide - Smart Notes

Cell Chemistry & Cell Components
Chemical Definitions
This section introduces fundamental chemical concepts essential for understanding cellular structure and function in anatomy and physiology.
Atom: The smallest unit of matter, composed of protons, neutrons, and electrons.
Matter: Anything that occupies space and has mass.
Isotope: Atoms of the same element with different numbers of neutrons.
Molecule: Two or more atoms chemically bonded together.
Compound: A substance formed from two or more different elements bonded together.
Subatomic Particles
Proton: Positive charge (+1)
Neutron: Neutral charge (0)
Electron: Negative charge (-1)
Bohr Model & Valence Shell
The Bohr model illustrates electrons orbiting the nucleus in energy levels.
The valence shell is the outermost electron shell, crucial for chemical bonding.
Isotopes
Isotopes are variants of elements with differing neutron numbers, affecting atomic mass but not chemical properties.
Definition: Atoms with the same number of protons but different numbers of neutrons.
Subatomic particle that differs: Neutron
Benefits: Used in medical imaging, dating fossils, and as tracers in biochemical research.
Chemical Bonding
Chemical bonds are forces holding atoms together, forming molecules and compounds.
Ionic Bonds: Formed by transfer of electrons; example: NaCl (table salt).
Covalent Bonds: Formed by sharing electrons.
Nonpolar: Equal sharing (e.g., O2).
Polar: Unequal sharing (e.g., H2O).
Hydrogen Bonds: Weak attractions between polar molecules, important in water and DNA structure.
Chemical Reactions
Chemical reactions involve breaking and forming bonds, essential for cellular metabolism.
Requirements: Reactants, energy, and enzymes.
Metabolism: All chemical reactions in the body.
Anabolism: Building complex molecules from simpler ones.
Catabolism: Breaking down complex molecules into simpler ones.
Energy: Capacity to do work; often released or absorbed in reactions.
Types of Chemical Reactions
Synthesis: (building molecules)
Decomposition: (breaking molecules)
Exchange: (swapping components)
Reversible: (can proceed in both directions)
Enzymes
Enzymes are biological catalysts that speed up chemical reactions by lowering activation energy.
Definition: Proteins that catalyze biochemical reactions.
Macromolecule type: Protein
Energy of Activation Curve: Enzymes lower the energy barrier for reactions, making them proceed faster.
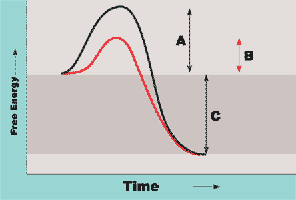
Inorganic vs Organic Compounds
Compounds in the body are classified as inorganic or organic based on their chemical structure.
Nutrients: Substances required for growth and maintenance.
Metabolites: Molecules involved in metabolism.
Inorganic compounds: Usually lack carbon (e.g., water, salts).
Organic compounds: Contain carbon and hydrogen (e.g., carbohydrates, proteins).
Example: Inorganic: NaCl; Organic: Glucose
Properties of Water
Water is vital for life due to its unique chemical properties.
Bond within molecule: Polar covalent bond
Bond between molecules: Hydrogen bond
Three properties:
High heat capacity
Solvent ability
Cohesion and adhesion
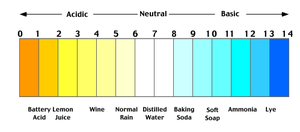
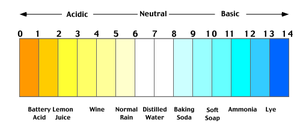
pH
pH measures the concentration of hydrogen ions in a solution, indicating its acidity or basicity.
Definition: pH is the negative logarithm of hydrogen ion concentration:
Acidic: pH < 7
Neutral: pH = 7
Basic: pH > 7
Blood pH: Approximately 7.4
Importance: pH affects enzyme activity and cellular function.
Example: Wine (acidic) has more hydrogen ions than baking soda (basic).
Macromolecules
Macromolecules are large biological molecules essential for cell structure and function.
Monomer: Single building block (e.g., amino acid)
Polymer: Chain of monomers (e.g., protein)
Conversion: Monomers join via dehydration synthesis; polymers break down via hydrolysis.
Macromolecule | Monomer | Polymer | Bonds | Function |
|---|---|---|---|---|
Carbohydrate | Monosaccharide | Polysaccharide | Glycosidic | Energy storage |
Protein | Amino acid | Polypeptide | Peptide | Structure, enzymes |
Nucleic Acid | Nucleotide | DNA/RNA | Phosphodiester | Genetic information |
Lipid | Fatty acid | Triglyceride | Ester | Energy storage, membranes |
Protein Structure
Four levels:
Primary (sequence)
Secondary (alpha helix, beta sheet)
Tertiary (3D folding)
Quaternary (multiple polypeptides)
Hemoglobin structure: Quaternary
Nucleic Acids
Three components of a nucleotide: Sugar, phosphate, nitrogenous base
DNA vs RNA: DNA is double-stranded, RNA is single-stranded
Genetic information: DNA
Protein production: RNA
High-energy bonds: ATP (a nucleotide)
Fats
Triglycerides
Phospholipids
Steroids
Fatty acids
Additional info: Academic context was added to expand brief points and clarify definitions, examples, and functions for exam preparation.
