 Back
BackChapter 2: The Chemical Level of Organization – Study Notes
Study Guide - Smart Notes

Atoms and Atomic Structure
Subatomic Particles
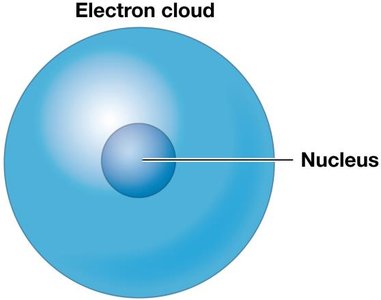
The atom is the smallest stable unit of matter, composed of three main subatomic particles: protons (positively charged), neutrons (neutral), and electrons (negatively charged). Protons and neutrons are similar in size and mass and are located within the nucleus at the center of the atom, while electrons are much lighter and orbit the nucleus in an electron cloud.
Protons (p+): Positive charge, found in the nucleus.
Neutrons (n): No charge, found in the nucleus.
Electrons (e-): Negative charge, orbit the nucleus.
Atomic Number and Structure
The atomic number is the number of protons in an atom and is unique for each element. Atoms typically have equal numbers of protons and electrons, making them electrically neutral (except for ions).
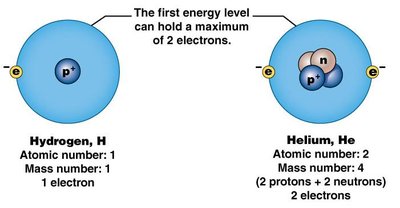
Example: Hydrogen (H) has an atomic number of 1, meaning it has 1 proton and 1 electron.
Electron Cloud and Shells
The electron cloud is a spherical area around the nucleus where electrons travel at high speed. Electron shells are two-dimensional representations of the electron cloud, with each shell holding a limited number of electrons. The electrical attraction between protons and electrons keeps electrons within their shells.
Elements and Isotopes
Elements
An element is a pure substance composed of atoms with the same number of protons. Each element has a unique atomic number and a chemical symbol (e.g., O for oxygen, Na for sodium).
Principal elements in the human body: Oxygen, carbon, hydrogen, nitrogen, calcium, phosphorus, potassium, sodium, chlorine, magnesium, sulfur, iron, iodine, and trace elements.
Element | % Body Weight | Significance |
|---|---|---|
Oxygen (O) | 65 | Component of water, essential for respiration |
Carbon (C) | 18.6 | Found in all organic molecules |
Hydrogen (H) | 9.7 | Component of water and most compounds |
Nitrogen (N) | 3.2 | Found in proteins, nucleic acids |
Calcium (Ca) | 1.8 | Bones, teeth, nerve impulses, muscle contraction |
Phosphorus (P) | 1.0 | Bones, teeth, nucleic acids |
Potassium (K) | 0.4 | Membrane function, nerve impulses |
Sodium (Na) | 0.2 | Blood volume, nerve impulses |
Chlorine (Cl) | 0.2 | Blood volume, water absorption |
Magnesium (Mg) | 0.06 | Cofactor for enzymes |
Sulfur (S) | 0.04 | Found in proteins |
Iron (Fe) | 0.007 | Oxygen transport |
Iodine (I) | 0.0002 | Thyroid hormones |
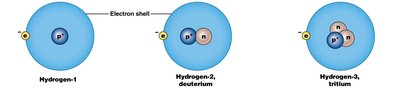
Isotopes and Radioisotopes
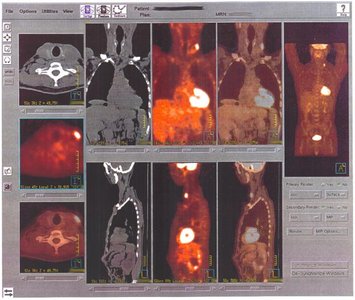
Isotopes are forms of the same element with different numbers of neutrons, resulting in different mass numbers but similar chemical properties. Radioisotopes have unstable nuclei and emit radiation as they decay, a process measured by half-life. Some radioisotopes are used in medical diagnostics, such as PET scans.
Electrons and Energy Levels
Electron Shells and Valence
Electrons occupy energy levels or shells around the nucleus. The first shell holds up to 2 electrons, while the second and third can each hold up to 8 electrons. The valence shell is the outermost shell, and its electron count determines an atom's chemical reactivity.
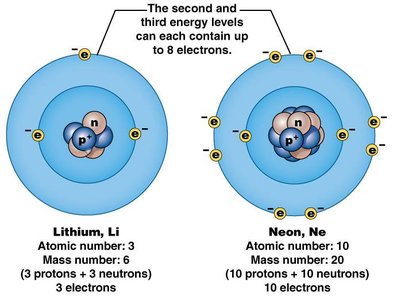
Atoms with unfilled valence shells are reactive and seek to fill them through chemical reactions.
Atoms with filled valence shells are stable (inert).
Molecules and Compounds
Definitions and Types
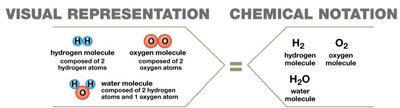
A molecule is formed when two or more atoms share electrons. A compound is a chemical substance composed of atoms of two or more different elements. Not all molecules are compounds (e.g., O2), and not all compounds are molecules (e.g., NaCl, which is held together by ionic bonds, not shared electrons).
Ions and Ionic Bonds
Ions
An ion is an atom or group of atoms with an electrical charge, formed by gaining or losing electrons. Cations are positively charged (loss of electrons), and anions are negatively charged (gain of electrons).
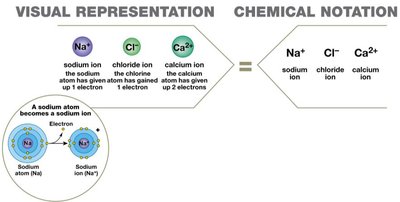
Ionic Bonds
Ionic bonds are formed by the attraction between cations and anions. One atom donates electrons (becoming a cation), and another accepts them (becoming an anion). The resulting oppositely charged ions attract, forming ionic compounds such as NaCl.
Covalent Bonds
Types of Covalent Bonds
Covalent bonds involve the sharing of electrons between atoms. They can be single (one pair shared), double (two pairs), or triple (three pairs). Nonpolar covalent bonds share electrons equally, while polar covalent bonds share electrons unequally, resulting in partial charges (e.g., water molecules).
Hydrogen Bonds
Hydrogen bonds are weak attractions between the partial positive charge of a hydrogen atom in a polar covalent bond and the partial negative charge of another atom (O, N, or F) in a different polar covalent bond. These bonds are important in water, proteins, and nucleic acids.
Chemical Reactions
Types of Chemical Reactions
Decomposition: Breaking a molecule into smaller fragments ()
Hydrolysis: Decomposition involving water ()
Synthesis: Assembling larger molecules from smaller ones ()
Dehydration Synthesis: Synthesis producing water ()
Exchange: Rearrangement of components ()
Reversible: Reactions that can proceed in both directions ()
pH and Homeostasis
pH measures the concentration of hydrogen ions in a solution. A pH of 7 is neutral, below 7 is acidic, and above 7 is basic (alkaline). The body tightly regulates pH, especially in blood (normal range: 7.35–7.45). Deviations can cause acidosis or alkalosis, affecting cellular function and survival.
Macromolecules
Types and Functions
Macromolecules are large, complex molecules essential for life. There are four main types:
Carbohydrates: Energy sources; monomer is monosaccharide (e.g., glucose).
Lipids: Energy storage, insulation, membrane structure; monomers include fatty acids and glycerol.
Proteins: Structure, movement, transport, enzymes, defense; monomer is amino acid.
Nucleic Acids: Store and process genetic information; monomer is nucleotide.
Carbohydrates
Monosaccharides: Simple sugars (e.g., glucose, fructose).
Disaccharides: Two monosaccharides joined (e.g., sucrose).
Polysaccharides: Many monosaccharides joined (e.g., starch, glycogen, cellulose).
Lipids
Fatty acids: Saturated (no double bonds) or unsaturated (one or more double bonds).
Glycerides: Fatty acids attached to glycerol (mono-, di-, triglycerides).
Steroids: Four-ring structure (e.g., cholesterol, hormones).
Phospholipids: Major component of cell membranes.
Proteins
Amino acids: 20 types, each with a central carbon, hydrogen, amino group, carboxyl group, and R group.
Peptide bonds: Link amino acids via dehydration synthesis.
Structure: Primary (sequence), secondary (alpha helix, beta sheet), tertiary (3D folding), quaternary (multiple polypeptides).
Enzymes: Catalysts that lower activation energy for reactions.
Nucleic Acids
DNA: Double helix, stores genetic information, bases A, T, C, G.
RNA: Single strand, involved in protein synthesis, bases A, U, C, G.
Nucleotides: Monomers with a pentose sugar, phosphate group, and nitrogenous base.
High-Energy Compounds
Cells use high-energy compounds, mainly derived from nucleotides, to store and transfer energy. Adenosine triphosphate (ATP) is the primary energy carrier, with energy released when its high-energy phosphate bonds are broken. ATPase is the enzyme that catalyzes ATP breakdown.
