Back
BackChapter 26: Fluid, Electrolyte, and Acid-Base Balance – Study Notes
Study Guide - Smart Notes

Fluid, Electrolyte, and Acid-Base Balance
Body Fluids and Homeostasis
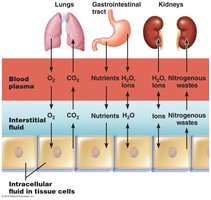
All body cells exist in a fluid environment, and essential exchanges (such as obtaining oxygen and nutrients, and excreting wastes) occur with the surrounding interstitial fluid (ISF). The ISF is continuous with blood plasma via exchange across permeable capillary walls. Normal cellular function depends on the physical and chemical homeostasis of body fluids; disturbances (e.g., dehydration) can be rapidly fatal.
Body Fluid Compartments
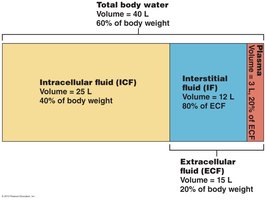
Total body water content is approximately 40 liters, which is about 60% of body weight. There are two major body fluid compartments:
Intracellular fluid (ICF): The water inside trillions of body cells (~2/3 of body water, 25 liters, 40% of body weight).
Extracellular fluid (ECF): The water outside the cells (~1/3 of body water, 15 liters, 20% of body weight), which includes:
Interstitial fluid (IF): ~12 liters, 80% of ECF
Blood plasma: ~3 liters, 20% of ECF
Body Fluid Composition
Water is the universal solvent of the body and contains a variety of solutes, classified as electrolytes and non-electrolytes:
Non-electrolytes: Molecules (usually organic) that do not dissociate into ions in solution (e.g., glucose, urea).
Electrolytes: Compounds that dissociate into charged ions in solution, conducting electrical current (e.g., inorganic salts, acids, bases).
Osmotic Activity and Osmolarity
Osmotic activity reflects the tendency of a solution to cause osmosis due to its osmolarity. Key definitions:
Molarity (M): Number of moles of solute per liter of solvent.
Osmolarity (Osm): Net number of solute particles per liter of solvent.
For example:
1M Glucose/L = 1 Osm/L
1M NaCl/L → 1M Na+ and 1M Cl- ions/L = 2 Osm/L
Body fluid osmolarity is approximately 300 mOsm.
Equivalence and Laboratory Values
Electrolyte concentrations are usually expressed in milliequivalents per liter (mEq/L), indicating the number of electrical charges per liter. For monovalent ions, 1 mmol = 1 mEq/L; for divalent ions, 1 mmol = 2 mEq/L. These values are commonly used in clinical laboratory tests such as the Basic Metabolic Panel (BMP) and Complete Metabolic Panel (CMP).

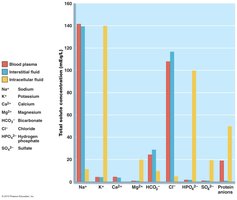
Composition of ECF and ICF
Body fluid compartments have distinct solute compositions:
Major ECF cation: Na+; major ECF anion: Cl-
Major ICF cation: K+; major ICF anions: HPO42- and proteins
This distribution is maintained by the Na+/K+ pump and the kidneys.
Fluid Exchange Between Compartments
Continual fluid exchange occurs between compartments, driven by osmotic and hydrostatic forces. Any change in osmolarity of a compartment leads to water movement (osmosis). Plasma-ISF exchange occurs at capillary beds, while ISF-ICF exchange occurs across cell membranes. Water always follows solute; changes in solute concentration cause osmosis and alter compartment fluid volumes.
Water Balance
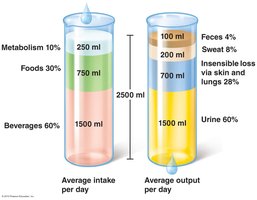
To maintain water balance, intake must equal output. Average water intake is about 2500 ml/day, from food, drink, and cellular metabolism. Water is lost via insensible loss (skin, lungs), perspiration, feces, and urine. The kidneys regulate water balance by adjusting urine production, primarily under the influence of antidiuretic hormone (ADH).
Overhydration and Types
Overhydration is a condition of positive water balance, typically when ECF gains volume. It can occur due to excessive intake of water, Na+, or both. Symptoms include nausea and muscle cramps; severe cases may lead to cerebral edema. Types include:
Isosmotic: Gain of water and Na+ (e.g., isotonic IV fluids); no major change in osmolality.
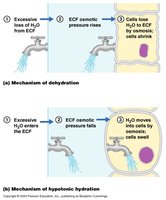
Hyposmotic: Gain of water only (e.g., SIADH); water shifts into ICF.
Hyperosmotic: Gain of Na+ more than water; water shifts out of ICF.
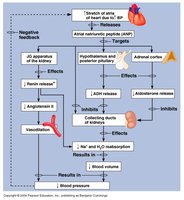
Atrial Natriuretic Peptide (ANP)
ANP is a peptide hormone released by atrial cardiac muscle fibers in response to overhydration (atrial stretch). It acts as a potent diuretic and natriuretic, antagonizing angiotensin II, increasing excretion of Na+ and water, and inhibiting aldosterone and ADH release. This leads to reduced blood volume and pressure.
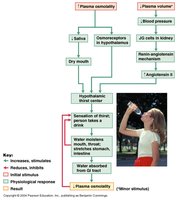
Regulation of Water Intake
Dehydration triggers the thirst mechanism, regulated by osmoreceptors in the hypothalamus. These receptors are stimulated when ECF becomes hypertonic or when blood pressure/volume drops. The hypothalamus signals the thirst center and triggers ADH release from the posterior pituitary, promoting water intake and retention.
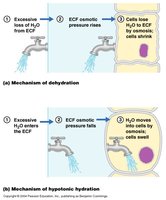
Clinical Dehydration and Types
Dehydration is a condition of negative water balance, typically when ECF volume falls. Causes include excessive loss of water, Na+, or both. Early signs include dry mouth, dry skin, oliguria, and hypotension. Types include:
Isosmotic: Loss of water and Na+ (e.g., hemorrhage, burns); no major change in osmolality.
Hyperosmotic: Loss of water only (e.g., diabetes, fever); water shifts from ICF to ECF.
Hyposmotic: Loss of Na+ more than water (e.g., Addison’s disease); water shifts into ICF.
Sodium Balance
Sodium plays a central role in fluid and electrolyte balance. Normal serum Na+ levels are 135-145 mEq/L. Sodium is the primary determinant of ECF volume and water distribution. Changes in plasma sodium levels affect ISF and ICF volumes due to osmosis.
Hypernatremia
Serum Na+ >145 mmol/L. Causes include decreased water intake, increased water loss, or renal issues (e.g., diabetes insipidus). Treatment involves adequate hydration.
Hyponatremia
Serum Na+ <135 mmol/L, usually secondary to overhydration. Causes include increased water intake, decreased GFR, or SIADH. Can lead to hyponatremic encephalopathy. Treatment includes fluid restriction and, if severe, hypertonic saline infusion.
Regulation of Potassium
Potassium is crucial for the function of excitable tissues. It is reabsorbed and secreted in the nephron as needed, regulated by aldosterone. Abnormalities (hypokalemia or hyperkalemia) can be rapidly fatal due to cardiac arrhythmias.
Hyperkalemia
Serum K+ >5.5 mmol/L. Causes include acidosis, renal failure, or aldosterone insufficiency. Treatments include calcium chloride, bicarbonate, insulin with glucose, diuretics, or dialysis.
Hypokalemia
Serum K+ <3.5 mmol/L. Causes include diarrhea, vomiting, hyperaldosteronism, or diuretic use. Treatment involves potassium replacement and addressing the underlying cause.
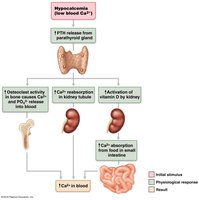
Regulation of Calcium
Calcium is vital for neuromuscular function, with 99% stored in bone. Homeostasis is mainly controlled by parathyroid hormone (PTH), which increases osteoclast activity, vitamin D3 activation, and renal Ca++ reabsorption. As Ca++ reabsorption increases, PO43- reabsorption decreases.
Hypercalcemia
Serum Ca++ >10.5 mg/dl. Symptoms include weak muscle contractions, arrhythmias, bone pain, constipation, and psychiatric effects. Causes include hyperparathyroidism and acidosis. Treatment includes diuretics, bisphosphonates, and calcitonin.
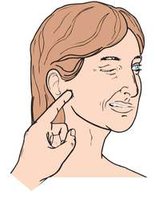
Hypocalcemia
Serum Ca++ <8.5 mg/dl. Symptoms include Chvostek’s sign (facial twitch) and Trousseau’s sign (carpal spasm). Causes include hypoparathyroidism, vitamin D deficiency, and alkalosis. Treated with calcium administration.

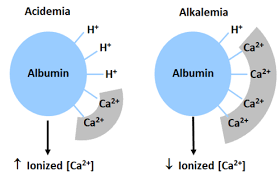
Relationship to Acidosis/Alkalosis
About 50% of serum Ca++ is bound to albumin; the rest is free and biologically active. In acidosis, more H+ binds to albumin, increasing free Ca++. In alkalosis, less H+ is bound, so more Ca++ binds to albumin, decreasing free Ca++.
Anion Regulation
Chloride (Cl-) is the major anion in the ECF, providing osmotic pressure along with Na+. Most Cl- is reabsorbed passively in the nephron, often coupled to Na+ transport. In acidosis, less Cl- is reabsorbed as more HCO3- is reabsorbed.
Acid-Base Balance
Regulation of body fluid pH is critical for protein and enzyme function. The normal blood pH range is 7.35–7.45. pH is defined as the negative logarithm of the hydrogen ion concentration:
Acidosis: arterial blood pH < 7.35; Alkalosis: arterial blood pH > 7.45.
Sources of Hydrogen Ions
Most H+ comes from metabolic acids (protein, lipid, and nucleotide metabolism, anaerobic respiration, and respiratory metabolism). Acid-base balance is regulated by chemical buffers (immediate), respiratory adjustments (seconds-minutes), and renal mechanisms (hours-days).
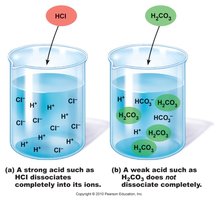
Acids and Bases
Acids: Proton donors; strong acids (e.g., HCl) dissociate completely, weak acids (e.g., H2CO3) dissociate partially.
Bases: Proton acceptors; strong bases dissociate completely, weak bases dissociate partially.