 Back
BackChapter 26: Fluid, Electrolyte, and Acid-Base Balance – Study Notes
Study Guide - Smart Notes

Fluid, Electrolyte, and Acid-Base Balance
Body Fluids
The human body is composed of a significant amount of water, which is distributed in various compartments. Understanding the distribution and regulation of body fluids is essential for maintaining homeostasis.
Total body water declines with age: Infants have the highest percentage (~73%), healthy males about 60%, healthy females about 50%, and elderly individuals about 45%.
The difference between males and females is due to higher body fat and lower skeletal muscle mass in females.
Fluid Compartments
Body fluids are distributed in two main compartments: intracellular and extracellular. Each compartment has distinct characteristics and functions.
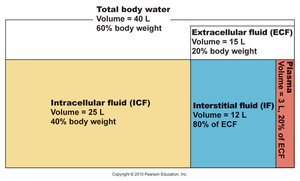
Intracellular fluid (ICF): Makes up about 2/3 of total body water (25 L), located within cells.
Extracellular fluid (ECF): Accounts for 1/3 of total body water (15 L), found outside cells. It is further divided into:
Plasma: 3 L, about 20% of ECF.
Interstitial fluid (IF): 12 L, about 80% of ECF, includes lymph, cerebrospinal fluid (CSF), synovial fluid, serous fluid, and gastrointestinal secretions.
Composition of Body Fluids
Water is the universal solvent in the body. Solutes are classified as electrolytes or nonelectrolytes, each with distinct properties and physiological roles.
Electrolytes: Dissociate into ions in water (e.g., NaCl, acids, bases, some proteins). They conduct electrical current and have high osmotic power, causing fluid shifts.
Nonelectrolytes: Mostly organic molecules (e.g., glucose, lipids, creatinine, urea) that do not dissociate in water and do not create charged particles.
Electrolyte Concentration in Body Fluids
The distribution of electrolytes varies between compartments and is crucial for physiological processes.
ECF: Major cation is Na+; major anion is Cl–. Plasma has higher protein and lower Cl– than IF.
ICF: Major cation is K+; major anion is HPO42–. Contains more soluble proteins than plasma.
Sodium and potassium concentrations are nearly opposite in ECF and ICF.
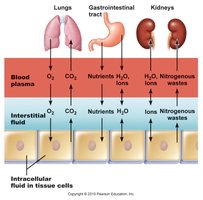
Fluid Movement Among Compartments
Osmotic and hydrostatic pressures regulate the continuous exchange and mixing of body fluids. Water moves freely along osmotic gradients, ensuring equal osmolality across compartments.
Changes in solute concentration in any compartment lead to net water flow.
Increased ECF osmolality causes water to leave cells; decreased ECF osmolality causes water to enter cells.
Water Balance and ECF Osmolality
Water Intake and Output
To maintain hydration, water intake must equal water output, averaging about 2500 ml/day in adults.
Water intake: 60% from beverages, 30% from food, 10% from metabolic water.
Water output: 60% via urine, 28% via insensible losses (skin and lungs), 8% via sweat, 4% via feces.

Maintenance of Body Fluid Osmolality
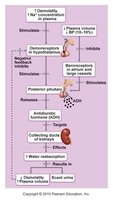
Osmolality is maintained at approximately 280–300 mOsm. Changes in osmolality regulate thirst and antidiuretic hormone (ADH) release.
Increased osmolality stimulates thirst and ADH release.
Decreased osmolality inhibits thirst and ADH release.
Regulation of Water Intake
The thirst mechanism, governed by the hypothalamic thirst center, is the primary driver for water intake.
Stimulated by increased plasma osmolality (1–2%), dry mouth, decreased blood volume or pressure, angiotensin II, or baroreceptor input.
Drinking water inhibits the thirst center via relief of dry mouth and activation of stretch receptors in the stomach and intestine.
Regulation of Water Output
Obligatory water losses occur through insensible loss (lungs and skin), feces, and urine. The kidneys play a key role in regulating urine volume to maintain homeostasis.
Minimum daily urine output is about 500 ml to excrete wastes.
Urine solute concentration and volume depend on fluid intake, diet, and other water losses.
Mechanisms and Consequences of ADH Release
ADH (antidiuretic hormone) regulates water reabsorption in the kidneys. Its release is controlled by hypothalamic osmoreceptors and is influenced by blood volume and pressure.
Low ADH: Dilute urine, decreased body fluid volume.
High ADH: Concentrated urine, increased body fluid volume.
Other triggers: Large changes in blood volume/pressure, intense sweating, vomiting, diarrhea, blood loss, burns, fever.
Disorders of Water Balance
Dehydration
Dehydration is a negative fluid balance resulting from excessive water loss. Causes include hemorrhage, burns, vomiting, diarrhea, sweating, water deprivation, diuretic abuse, and endocrine disturbances.
Signs: Thirst, dry mouth, flushed skin, low urine output (oliguria).
Consequences: Weight loss, fever, confusion, hypovolemic shock, electrolyte loss.
Hypotonic Hydration
Also known as water intoxication, this condition results from excessive water intake or renal insufficiency, leading to low ECF osmolality and cellular swelling.
Consequences: Nausea, vomiting, muscle cramps, cerebral edema, possible death.
Treatment: Hypertonic saline administration.
Edema
Edema is the atypical accumulation of interstitial fluid, causing tissue swelling (not cellular swelling).
Causes: Increased capillary hydrostatic pressure or permeability, hypoproteinemia, lymphatic blockage.
Consequences: Low blood pressure, impaired circulation.
Electrolyte Balance
Overview of Electrolyte Balance
Electrolytes include salts, acids, bases, and some proteins. Electrolyte balance primarily refers to salt balance, which is vital for fluid movement, excitability, secretory activity, and membrane permeability.
Salts are ingested and produced metabolically; lost via perspiration, feces, urine, and vomiting.
Central Role of Sodium
Sodium is the most abundant cation in the ECF and is crucial for controlling ECF volume, water distribution, and blood pressure.
Sodium salts contribute 280 mOsm of the total 300 mOsm ECF solute concentration.
Only cation exerting significant osmotic pressure.
Changes in Na+ levels affect plasma volume, blood pressure, and ECF/IF volumes.
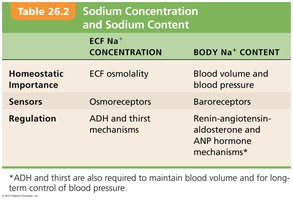
Sodium Concentration vs. Sodium Content
The concentration and total content of sodium have distinct physiological implications and regulatory mechanisms.
ECF Na+ Concentration | Body Na+ Content | |
|---|---|---|
Homeostatic Importance | ECF osmolality | Blood volume and blood pressure |
Sensors | Osmoreceptors | Baroreceptors |
Regulation | ADH and thirst mechanisms | Renin-angiotensin-aldosterone and ANP hormone mechanisms* |
*ADH and thirst are also required to maintain blood volume and for long-term control of blood pressure.
Regulation of Sodium Balance
Sodium balance is closely linked to blood pressure and volume regulation. There are no direct sodium receptors; instead, neural and hormonal mechanisms respond to changes in blood pressure or volume.
Aldosterone: Increases Na+ reabsorption in the kidneys, decreasing urinary output and increasing blood volume. Triggered by the renin-angiotensin-aldosterone mechanism and high K+ levels.
ANP (Atrial Natriuretic Peptide): Released in response to atrial stretch (high blood pressure), decreases blood pressure and volume by inhibiting ADH, renin, and aldosterone, and increasing Na+ and water excretion.
Other hormones: Estrogens increase NaCl reabsorption; progesterone decreases it; glucocorticoids increase Na+ reabsorption and promote edema.
Cardiovascular baroreceptors: Detect changes in blood volume/pressure and adjust sympathetic impulses to the kidneys, affecting GFR and Na+ output.
Regulation of Potassium Balance
Importance of Potassium
Potassium is essential for maintaining resting membrane potential (RMP) in neurons and muscle cells, especially cardiac muscle.
High ECF [K+]: Decreases RMP, causing depolarization and reduced excitability.
Low ECF [K+]: Causes hyperpolarization and nonresponsiveness.
Hyperkalemia (high K+) and hypokalemia (low K+) can disrupt heart function and cause sudden death.
Potassium and Acid-Base Balance
Potassium participates in the body's buffer system. H+ shifts in and out of cells in the opposite direction of K+ to maintain cation balance.
ECF K+ rises with acidosis and falls with alkalosis, affecting excitable cells.
Regulation of Potassium Balance
Potassium balance is primarily controlled by the kidneys, specifically in the cortical collecting ducts.
High K+ in ECF increases secretion by principal cells.
Low K+ reduces secretion; type A intercalated cells reabsorb K+.
Aldosterone stimulates K+ secretion and Na+ reabsorption; adrenal cortical cells are sensitive to ECF K+ levels.
Abnormal aldosterone levels can severely affect K+ balance.
