Back
BackCHAPTER 2
Study Guide - Smart Notes

Chemistry of Life
Introduction
This chapter provides foundational chemistry concepts essential for understanding anatomy and physiology. It covers the nature of matter, atomic structure, chemical bonds, reactions, and the properties of water and organic molecules, all of which are critical for biological systems.
Basic Chemical Concepts
Matter, Elements, Atoms, and Ions
Matter: Anything that occupies space and has mass.
Element: A pure substance consisting of one type of atom, distinguished by its atomic number (number of protons).
Atom: The smallest unit of an element that retains its properties; cannot be broken down by ordinary chemical means.
Ion: An atom or molecule that has gained or lost electrons, resulting in a net charge. Cations are positively charged; anions are negatively charged.
Atomic Structure and Related Terms
Atomic Nucleus: The dense center of an atom, containing protons and neutrons.
Proton: Positively charged particle in the nucleus.
Neutron: Electrically neutral particle in the nucleus.
Electron: Negatively charged particle orbiting the nucleus in energy levels (shells).
Atomic Weight (Atomic Mass): The sum of protons and neutrons in an atom.
Valence: The number of electrons in the outermost shell, important for chemical bonding.
Isotope: Atoms of the same element with different numbers of neutrons. Some isotopes are radioactive, releasing energy from the nucleus.
Chemical Bonds
Types and Strengths of Chemical Bonds
Covalent Bonds: Atoms share electrons. Nonpolar covalent bonds share electrons equally (e.g., carbon backbones), while polar covalent bonds share electrons unequally (e.g., water).
Ionic Bonds: Electrons are transferred from one atom to another, forming charged ions (e.g., NaCl). Ionic compounds often dissociate in water.
Hydrogen Bonds: Weak attractions between a slightly positive hydrogen and a slightly negative atom (often oxygen or nitrogen). Important in stabilizing large molecules like DNA and proteins.
Van der Waals Forces: Weak, transient attractions due to temporary charge differences; important for molecular specificity and selectivity.
Bond Strength Ranking: Covalent > Ionic > Hydrogen > Van der Waals
Chemical Reactions
Types of Chemical Reactions
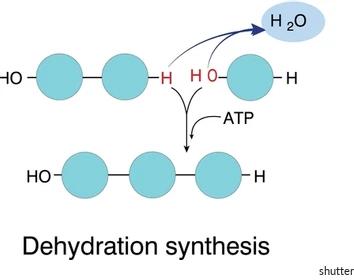
Dehydration Synthesis: Two molecules combine to form a larger molecule, releasing water. This is an endergonic (energy-requiring) process.
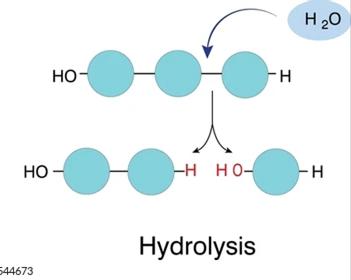
Hydrolysis: Water is added to split a large molecule into smaller components. This is an exergonic (energy-releasing) process.
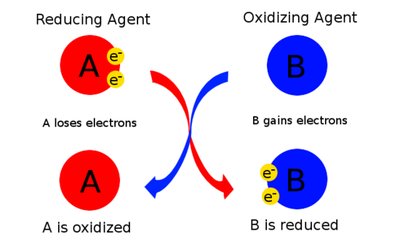
Redox (Reduction-Oxidation) Reactions: Involve the transfer of electrons. Oxidation is the loss of electrons; reduction is the gain of electrons. These reactions are always coupled.
Exchange Reactions: Atoms or groups of atoms are exchanged between molecules, forming new products.
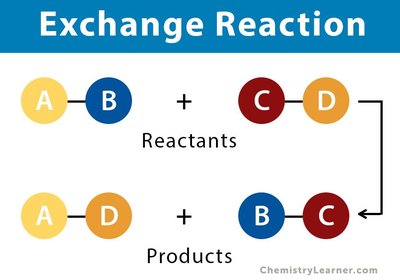
Chemical Notation
Rules of Chemical Notation
Reactants are substances entering a reaction; products are substances formed.
Arrows indicate the direction of the reaction; double arrows indicate reversibility.
Subscripts show the number of atoms in a molecule; coefficients show the number of molecules.
Superscripts (+ or -) indicate ionic charge.
Chemical reactions rearrange atoms but do not create or destroy them.

Acid-Base Balance and the pH Scale
Acids, Bases, and pH
Acids: Substances that release hydrogen ions (H+) in solution.
Bases: Substances that release hydroxide ions (OH-) in solution.
pH Scale: Measures hydrogen ion concentration from 0 (most acidic) to 14 (most basic); 7 is neutral (pure water).
Each pH unit represents a tenfold change in H+ concentration.
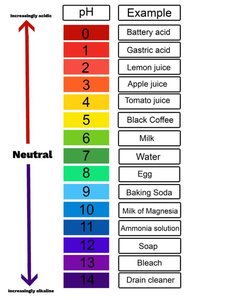
Properties of Water and Solutions
Unique Properties of Water
Exists in three states: liquid, solid, gas.
High heat capacity and cohesive properties (surface tension, capillary action).
Universal solvent—dissolves many substances due to polarity.

Solutions and Concentrations
Solvent: The liquid in which substances dissolve (water is the most common biological solvent).
Solute: The substance dissolved in a solvent.
Solution: A homogeneous mixture of solvent and solute.
Hypertonic: Higher solute concentration outside the cell; water leaves the cell, causing shrinkage.
Hypotonic: Lower solute concentration outside the cell; water enters the cell, causing swelling or bursting.
Isotonic: Equal solute concentration inside and outside the cell; no net water movement.
Hydrophilic: Water-loving, dissolves in water.
Hydrophobic: Water-fearing, does not dissolve in water.
Organic Molecules
General Properties
All contain carbon and hydrogen atoms, often arranged in backbones or rings.
Major classes: carbohydrates, proteins, lipids, nucleic acids.
Formed from specific monomers (building blocks).
Carbohydrates
Structure and Function
Monomers: Monosaccharides (e.g., glucose, fructose).
Disaccharides: Two monosaccharides joined (e.g., sucrose, lactose).
Polysaccharides: Long chains of monosaccharides (e.g., cellulose, starch).
Function: Primary energy source and structural component in cells.

Proteins
Amino Acids, Peptides, and Protein Structure
Monomers: Amino acids (20 types), each with an amine group, carboxyl group, and variable R group.
Peptides: Short chains of amino acids (dipeptide, tripeptide, polypeptide).
Protein Structure:
Primary: Sequence of amino acids.
Secondary: Alpha-helix or beta-sheet folding.
Tertiary: 3D globular shape, stabilized by various bonds.
Quaternary: Multiple polypeptide chains forming a functional unit (e.g., hemoglobin).
Function: Structural support, enzymes, hormones, receptors, immune defense.
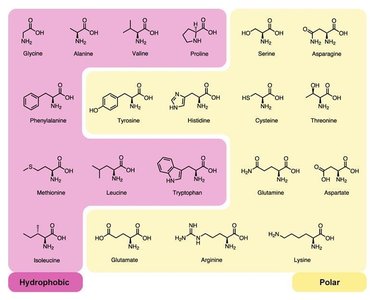
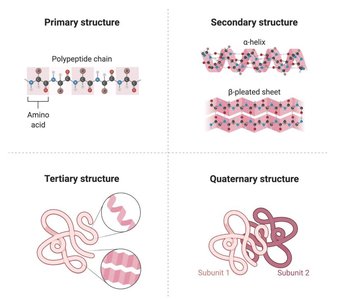
Lipids
Types and Functions
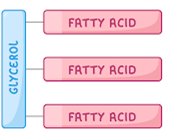
Triglycerides: Glycerol + 3 fatty acids; energy storage.
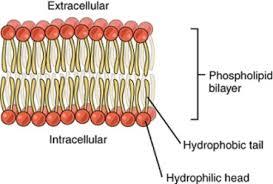
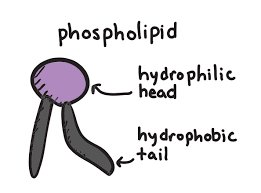
Phospholipids: Glycerol, 2 fatty acids, and a phosphate group; main component of cell membranes (hydrophilic head, hydrophobic tail).
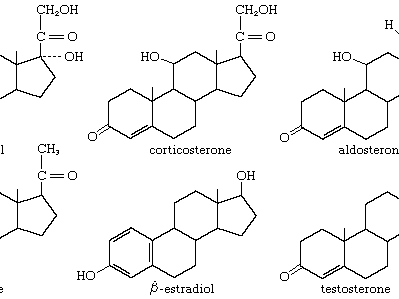
Steroids: Four fused carbon rings; include cholesterol, sex hormones, and corticosteroids.
Prostaglandins: Fatty acid derivatives with hormone-like effects.
Cholesterol: Precursor for vitamin D and steroid hormones; important for membrane structure.
Nucleic Acids
Structure and Function
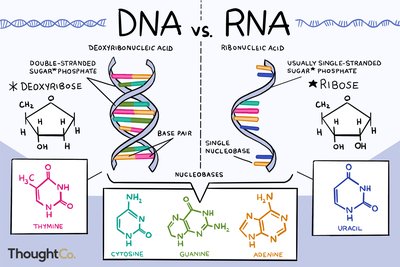
Monomers: Nucleotides (pentose sugar, phosphate group, nitrogenous base).
Nitrogenous Bases: Purines (adenine, guanine), Pyrimidines (cytosine, thymine, uracil).
Function: Store and transmit genetic information, direct protein synthesis, energy transfer (ATP).
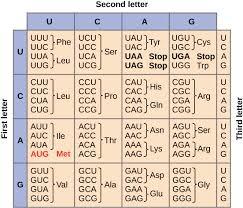
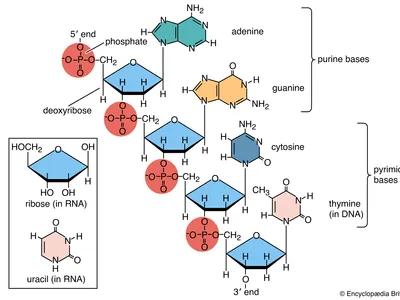
DNA and RNA
DNA (Deoxyribonucleic Acid): Double helix, deoxyribose sugar, bases A-T and G-C; stores genetic information.
RNA (Ribonucleic Acid): Single-stranded, ribose sugar, bases A-U and G-C; involved in protein synthesis (mRNA, tRNA, rRNA).
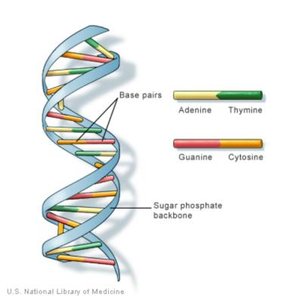
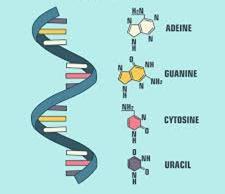
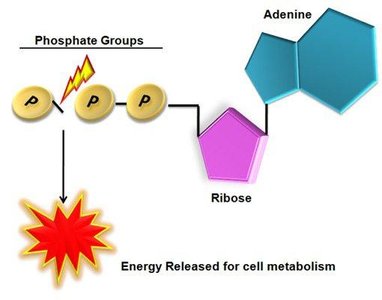
Adenosine Triphosphate (ATP)
ATP is the primary energy carrier in cells.
Energy is stored in high-energy phosphate bonds and released during hydrolysis to ADP or AMP.
ATP can be regenerated from ADP and AMP through cellular respiration or photosynthesis.